Canine Chronic Hepatitis
Chavezlloyd Alexandria Baldago Chronic hepatitis is a condition in dogs that may occur due to various disease processes. It indicates a previous occurrence of inflammation and possibly cell death in the liver. The inflammation is caused by the infiltration of different types of white blood cells that are involved in the immune system. Necrosis, which refers to the death of a large number of liver cells, may also occur. The invasion of white blood cells and cell death in a dog’s liver can be due to previous damage caused by infectious agents like viruses or bacteria or as a result of toxic damage. Toxic damage may occur due to poisons ingested by the dog or abnormal accumulation of substances required by the body, such as copper. Though some breeds like Bedlington Terriers, Labrador Retrievers, Doberman Pinschers, Dalmatians, West Highland White Terriers, Welsh Corgis, Keeshonds, and others are notably affected, this toxicity can affect any breed of dog. Copper accumulation in hepatocytes can cause oxidative damage, and its presence can escalate hepatocyte damage caused by other factors. Until dietary recommendations are modified and implemented, this problem will continue to affect canines. Inflammation and cell death can also occur due to a primary attack by the immune system against liver cells, known as an “autoimmune” disease. Cancer in the liver is not called chronic hepatitis, even if it causes similar damage. Chronic vs Acute The term “chronic” refers to a condition that has been causing damage for a prolonged period, typically lasting several weeks or more. On the other hand, “acute” hepatitis is usually characterized by a shorter duration of just a few days. While some cases of acute hepatitis can be treated successfully, many types of chronic hepatitis are not curable. However, with appropriate treatment and close monitoring, a significant number of patients with chronic hepatitis can maintain a good quality of life with minimal clinical symptoms for an extended period. Chronic hepatitis can affect any breed of dog regardless of gender or age, although it is more common in middle-aged or older dogs. Certain breeds may be more prone to specific types of hepatitis. For instance, some breeds may develop chronic hepatitis due to the accumulation of copper in the liver cells. The excessive concentration of copper harms the liver cells and, if left untreated, typically leads to severe chronic hepatitis. Symptoms The liver has multiple functions, therefore the clinical signs associated with liver disease can vary significantly. Symptoms of hepatitis in dogs can include: Lack of appetite Lethargy Vomiting Increased urination Excessive thirst and urination Swollen belly filled with fluid (ascites) Yellowish gums (jaundiced) and moist tissues Seizures, mental dullness Diagnosis Apart from obtaining a detailed history of the affected dog’s health prior to the appearance of symptoms, the next step would be to conduct a comprehensive physical examination of the dog, which includes carrying out a blood chemical profile, a complete blood count, an electrolyte panel, and a urinalysis. The results of the bloodwork will enable your veterinarian to identify any signs of impaired kidney function. To further evaluate the disease, complete abdominal ultrasonography is essential. This diagnostic tool screens for concurrent diseases and helps acquire bile. It’s important to note that the liver can appear normal upon examination, and changes seen in chronic hepatitis can include uniform increases in liver echogenicity, decreased distinction of portal vein margins, and normal to small liver size. Abnormalities in the liver parenchyma, biliary tree, portal vein, and peritoneum should be assessed, along with acquired shunting and the presence of free peritoneal fluid. In order to accurately diagnose chronic hepatitis, a liver biopsy is necessary. There are 3 ways to perform a liver biopsy, surgically, via laparoscopy, or through the skin using a special needle under ultrasound guidance. However, surgical or laparoscopic biopsies are more informative compared to ultrasound-guided biopsies. The information obtained from the biopsy is required to determine the type and severity of liver disease, as well as to assess your dog’s prognosis. Treatment The treatment for chronic hepatitis in dogs is a complex process that depends on the severity and type of liver disease, as well as the clinical signs exhibited by the dog. In severe cases, hospitalization, intravenous fluid therapy, and supportive care may be required. The most common medications prescribed for treating the disease are immunosuppressive or anti-inflammatory. In some cases, dietary changes may be necessary. Certain medications may also be used in specific situations, such as dogs whose illness is linked to copper accumulation, abdominal fluid build-up, or neurological symptoms. Life Expectancy Although this condition cannot be cured with the available treatments, the good news is that the dog can still live a good quality of life for months and even years with continued therapy. Regular blood work is necessary to ensure the dog is responding well to the treatment. This helps to adjust the medication and keep the dog relatively free of clinical signs. References: https://bluepearlvet.com/medical-articles-for-pet-owners/chronic-hepatitis-in-dogs/ https://todaysveterinarypractice.com/hepatology/canine-chronic-hepatitis-diagnosis-treatment/ https://www.msdvetmanual.com/digestive-system/hepatic-diseases-of-small-animals/canine-chronic-hepatitis#:~:text=Canine%20chronic%20hepatitis%20is%20a,be%20predisposed%20to%20chronic%20hepatitis.
Respiratory Tract Disease Complex in Cats

Sushant Sadotra, PhD/Diagnostic specialist Feline respiratory disease (FRD) syndrome or feline upper respiratory tract disease complex is a common infection in cats caused mainly by Feline Herpesvirus (FHV-1), Feline Calicivirus (FCV), Chlamydophila felis, Mycoplasma spp., and Bordetella bronchiseptica. About 90% of all upper respiratory infections are caused by FHV-1 and FCV. Common Symptoms: · Sneezing · Nasal congestion · Conjunctivitis (inflammation of the membranes lining the eyelids) · Discharge from the nose or eyes (clear, purulent, or cloudy containing pus). · Difficulty breathing · Ulcers in the mouth Less specific symptom · Less appetite · Lethargy · Fever · Enlarged lymph nodes · Blepharospasm (squinting) Sources of infection: · Susceptible cats can get an infection by contagious particles in saliva or secretions from the nose or eyes shredded by an infected cat. · Most cases are associated with direct contact · Natural transmissions can also occur via aerosol droplets. Stress may also cause a secondary course of illness. Real-Time PCR for Diagnosis: A definitive diagnosis is based on clinical signs and laboratory testing for the isolation and identification of the infection. The Polymerase chain reaction (PCR) test is one of the sensitive tests and most reliable for detecting the presence of infectious agents. PCR detects the genomic material of the pathogen and determines its presence in the host. It is often more sensitive and specific than other available tests. However, false negative results are still expected. In the case of patients with latent herpes infections where the FHV-1 is found in the trigeminal ganglion can give negative PCR results. In the case of Chlamydophila, 2-3 days of antibiotic treatment for patients can also have negative PCR results. Samples of ocular, nasal, or caudal pharyngeal secretions for PCR assay are best for the diagnosis and identification of causative agents. Sample collection tips: · Ocular: Moisten with tears/exudate well or firmly swab both of the conjunctival sacs with a sterile swab. · Clinical lesions: Prefer to swab from the nasal and pharyngeal areas. After sample collection, place the swab into the preservation buffer and mix thoroughly. If not for immediately use, please keep the mixture at 4°C (no more than 3 days) until nucleic acid extraction.
Feline Herpesvirus Infection- Diagnosis

Trinh Mai Nguyen Tang Feline herpesvirus-1 (FHV-1) is a feline respiratory infection virus also known as feline viral rhinotracheitis (FVR) [1]. The Herpes virus was first isolated by scientists Crandell and Maurer in 1958 in cats with respiratory infections [2]. This virus has a prominent genome with large double stranded DNA, belonging to the family Herpesviridae [3]. This virus is characterized by cat-to-cat transmission with an exposure rate of up to 97% [4]. Herpes virus can be inactivated at 37oC around 3 hours or at 56oC in 5 mins. Meanwhile, the virus can remains infective in the enviroment approximately 5 months and a month at 25oC [5]. Once a cat is infected with the herpes virus, it is incredibly difficult to completely treat it since the virus can enter a dormant state and continue to survive in the cat for the remainder of its life [6]. Cats are not infectious during this latent period, but if they are sick or going through a stressful period, the virus may be reactivate. If this occurs, the cat will once more get the infection and may represent symptoms [7]. Herpes viruses can be latent in the ganglion, attach to sensory nerves and reach nerve cells, persist in the nucleus of infected nerve cells and do not replicate, leading to the process of detecting this virus becomes difficult [7-8]. As reported by Ngoc.N.T and her colleagues, herpes virus can infect cats of any age, however, kittens are more susceptible [9]. Specifically, the prevalence of virus infection in cats younger than 6 months old, 6-12 months old and over 12 months old were 52.17%, 33.33% and 19.05%, respectively [9]. Although previous reports have demonstrate that gender has no effect on the incidence of herpes virus infection [7-8], but Henzel et al. (2002) found that isolates from female cats are substantially taller than isolates from male cats [10]. Clinical Symptoms Herpes virus enters the cat’s body by contact with infected tears, nose, saliva, or items, and then multiplies rapidly in the epithelium of the nose, nasopharynx, and conjunctival mucosa leading to primary infection [11]. In cats infected with FHV-1, signs of sadness, moodiness, lethargy, sneezing, fever, and discharge from the eyes and nose have been noted (figure 1-A), a process that is frequently extended 2-4 days or longer, depending on the immunological system of the cat [11]. Secondary infection occurred after the fourth day of incubation, with symptoms of infection in the throat, bronchi, and bronchioles, and the nasal and conjunctival epithelium necrosing [12]. Conjunctivitis is a common herpes virus symptom, indicated by congestive and exudative symptoms that develop over many days to purulent discharge (figure 1-B) [13]. Gaskell and Dawson (1988) found lung infection or bronchitis in cats, with kittens dying from pneumonia at a greater incidence than adult cats [11,14]. Some other atypical symptoms such as mouth and skin ulcers, dermatitis or neurological signs are rarely seen [7]. Furthermore, the mean white blood cell (WBC) count of cats infected with FHV-1 (17.77 ± 0.70 x103/μl) was slightly increased compared with that of normal cats (4.6-12.8 x 103/μl) [9], in which neutrophils, eosinophils and monocytes all showed signs of slight increase compared. Secondary infections of the eyes, upper respiratory tract, and necrotic ulcers of the mouth can all cause high white blood cell counts [11]. Table 1 displays the white blood cell count. Table 1. Hematological results of cats was infected with FHV-1 [9]. Targets Unit Reference ± SE Red blood cells ´ 106/μl 7-10,7 10,20 ± 0,64 Hemoglobin content g/dl 11,3-15,5 13,47 ± 0,54 RBC mass % 33-45 38,78 ± 1,41 Average volume of red blood cells fl 41-49 45,16 ± 0,75 Average amount of hemoglobin in red blood cells pg 14-17 15,57 ± 0,24 Platelet count ´ 103/μl 180-680 362,50 ± 30,82 WBC count ´ 103/μl 4,6-12,8 17,77 ± 0,70 Lymphocytes ´ 103/μl 1,05-6,00 4,50 ± 0,36 Mono leukocytes ´ 103/μl 0,05-0,68 0,96 ± 0,13 Neutrophils polymorphonuclear leukocytes ´ 103/μl 2,32-10,01 11,47 ± 0,45 Eosinophils ´ 103/μl 0,1-0,6 0,78 ± 0,08 Basophils ´ 103/μl 0-0,14 0,07 ± 0,01 Laboratory Diagnosis In the laboratory, there are many different methods used to determine FHV. Common approaches include PCR, virus isolation in cell culture, and indirect fluorescent antibody staining of tissue samples for viral antibody detection [8,16]. PCR FHV is one of the most common causes of upper respiratory tract illness in cats. Infected cats would show upper respiratory signs. Co-infection of FHV with other pathogens makes the clinical signs more severe, particularly feline calicivirus, Chlamydophila felis, Bordetella pneumoniaseptica, Mycoplasma species, Staphylococcus spp., or Escherichia coli [6]. When a cat is suspected of having a viral infection, a swab can be used to collect nasal, ocular, oropharyngeal secretions, corneal debris, aqueous humor, corneal samples, blood, or biopsies. By amplifying viral DNA, tPCR can detect genetic material of FHV in specimen. However, if the cat is not in the infectious phase, no virus particles will be shed, rendering the PCR test ineffective [19-20]. ELISA method – detecting IgG antibodies The enzyme-linked immunosorbent test (ELISA) is used to determine IgG antibodies against FHV or FHV-1 utilizing serum, aqueous humor, and cerebrospinal fluid samples [15]. This approach, however, cannot discriminate between diseased and vaccinated cats. Due to the extended latent period of FHV, the cat’s body will generate antibodies to combat it [8]. These neutralizing antibodies manifest 20-30 days after the first infection. As a result, the presence of antibodies in the serum signals a prior infection but does not always correspond with clinical signs [8]. Immunofluorescent antibody assay Another approach for detecting FHV is immunofluorescence antibody (IFA) testing on corneal or conjunctival smears or biopsiengs. Through an antigen-antibody response, this assay may identify viral proteins produced in cells. However, this approach is thought to be less sensitive than viral isolation or PCR [16]. Virus isolation This is a traditional method that can detect viruses through isolation of conjunctival debris, nose, oropharynx, or postmortem lung samples from infected cats [8]. This traditional method can detect
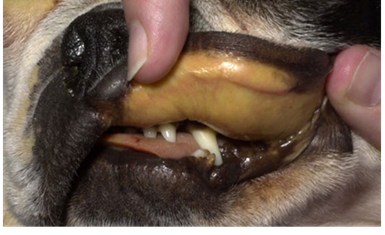
Canine Lyme Disease
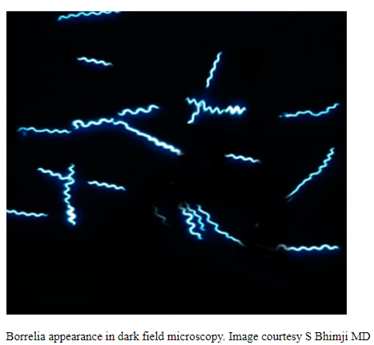
Oliver Organista, LA Lyme disease is a disease caused by the bacterium Borellia burdorgferi; a worm like, spiral-shape bacterium of spirochete class in the genus Borellia. The bacterium B. burgdorferi is transmitted through a bite of infected blacklegged tick or deer tick (Ixodes scapularis) to dogs and humans[1]. Different life-stage of I. scapularis ticks emerge at different times of the year (varies according to geographic location), giving a seasonality to Lyme disease transmission dynamics. It appears primarily in specific areas including the southern New England states; eastern Mid-Atlantic states; the upper Midwest, particularly Wisconsin and Minnesota; and on the West Coast, particularly northern California in the United States. It is also present in Europe and Asia[7]. Most of the areas where to find them are in forest or grassy, wooded, marshy areas near rivers, lakes or ocean, and are common in homes and buildings in secluded or rural areas. In Canada, there 2 types of blacklegged or deer tick that can spread Lyme disease. The blacklegged tick (Ixodes scapularis) and the blacklegged (Ixodes pacificus) [3] . Dogs tend to be bitten by infected I. scapularis adults, which are most active in the cooler early spring and late fall months [2]. An adult female tick is rarely (if ever) transmitted the B. burgdorferi to her offspring. Ticks most commonly become infected as juveniles after a bloodmeal on an infected wildlife host (most commonly rodents). Because ticks typically feed only one time per life stage, the next opportunity for B. burgdorferi transmission is during the next bloodmeal in the tick’s next life stage[2]. Typically, Lyme disease symptoms will take a couple of months or more to appear (2-5 months) after getting infected [8]. Symptomatically, Lyme disease can be difficult to distinguish from anaplasmosis because the signs of the diseases are very similar, and they occur in essentially the same areas of the country. Lyme disease is diagnosed through a blood test that shows whether an animal has been exposed to the bacterium[11]. Common symptoms that will appear are: Lameness: An inability to use one or more limbs is one of the most common symptoms of Lyme disease in dogs. Swollen lymph nodes: found in the neck, chest, armpits, groin, and behind the knees, are typically the first to show swelling. Lymph node swelling indicates an immune response triggered to fight the disease. Joint swelling: Swollen joints, stiff walking, or avoidance to touch may be other signs of the disease. Fatigue: Dogs with Lyme disease may also exhibit flu-like symptoms of low energy and lethargy. Loss of appetite: Losing interest in eating, especially if it leads to weight loss, is another sign that a dog may have Lyme. Fever: In addition to the above symptoms, a dog may have a fever caused by the Lyme disease infection. In rare cases, if Lyme disease is left untreated it can lead to damage in the kidneys, nervous system, and heart. Lyme disease affecting the kidneys is the second most common syndrome in dogs and is generally fatal. Facial paralysis and seizure disorders have been reported in the disease form affecting the nervous system. The form of the disease that affects the heart is rare. [10]. The most commonly used to diagnose Lyme disease in dogs are the serologic assays. Although some laboratories still use traditional serologic methods (e.g., whole-cell enzyme-linked immunosorbent assay and immunofluorescence assay), these assays have largely been replaced by serologic assays that detect host antibodies to specific B. burgdorferi proteins. These assays are qualitative, providing a yes/no answer regarding B. burgdorferi serostatus[3]. Treatment is generally recommended for seropositive dogs that display clinical signs of Lyme disease or are asymptomatic but have evidence of protein-losing nephropathy[4]. Most frequently antibiotics used to treat Lyme disease in dogs are doxyclycline and monicycline, at a dosage of 10mg/kg PO q12h to q24h for 30 days [2][5]. Amoxicillin and erythromycin are other antibiotics that can be used for treating the disease. A non-steroidal anti-inflammatory (carprofen or deracoxib) may also be given to the patient [6]. A possible complications may occur when treating Lyme disease. Some dogs who take antibiotics can develop loss of appetite, vomiting and diarrhea. Once infected, a dog will always have the bacteria that cause Lyme disease in his or her body. Therefore, relapses are possible; lookout for unexplained fever, swollen lymph nodes, and/or lameness. A small percentage of dogs develop kidney failure as a result of Lyme disease. Clinical signs include vomiting, weight loss, poor appetite, lethargy, increased thirst and urination, and abnormal accumulations of fluid within the body. [6] The best way to protect from Lyme disease is to use tick-preventive products year-round. Several safe and effective commercial parasiticides are available for tick control on dogs and cats, including systemics (isoxazolines), topicals (permethrin, fipronil), and collars. Another effective strategy is vaccination. Other prevention strategies include reducing exposure to ticks and avoiding areas with ticks [2]. References [1] Lyme Disease Diagnostic Market – Growth, Trends, COVID-19 Impact, and Forecasts (2022 – 2027), MOdor Intelligence, January 202 [2] Lyme Disease in Dogs: Signs and Prevention, Kathryn E. Reif, MSPH, PhD.,April 2020, https://todaysveterinarypractice.com/parasitology/lyme-disease/ [3] Lyme Disease, IPAC (https://ipac-canada.org/lyme-disease.php) [4] / Littman MP, Gerber B, Goldstein RE, et al. ACVIM consensus update on Lyme borreliosis in dogs and cats. J Vet Intern Med 2018;32(3):887-903. [5] Mullegger RR. Dermatological manifestations of Lyme borreliosis. Eur J Dermatol. 2004 Sep-Oct;14(5):296-309. PMID: 15358567 [6] How to Treat Lyme Disease in Dogs, Jennifer Coates, DVM, December 2014, PETMD (https://www.petmd.com) [7] Lebech AM. Polymerase chain reaction in diagnosis of Borrelia burgdorferi infections and studies on taxonomic classification. APMIS Suppl. 2002;(105):1-40. PMID: 11985118 [8] Could Your Dog Have Lyme Disease? How to Recognize the Symptoms and Get Treatment, Lavanya Sunkara , July 2022, GoodRx Health [9] Everything You Need To Know About Lyme Disease In Dogs, Kimberly Alt, July 2022, Canine Journal [10] Lyme Disease (Lyme Borreliosis) in Dogs, Reinhard K. Straubinger, DrMedVetHabil, PhD, Institute for Infectious Diseases and Zoonoses, Department of Veterinary Sciences, Faculty of Veterinary Medicine, LMU, October 2022 [11] Lyme disease: A pet owner’s guide, American Veterinary Medical Association
Psittacine Beak and Feather Disease

Long Pham Introduction Psittacine beak and feather disease (PBFD) is an infectious viral disease that infects psittacine birds. This disease affects Old World (Australian and African) psittacine birds and New World (Americas) psittacine birds (Greenacre, 2005). The peracute and acute form of this disease can cause sudden death, while the chronic form of this disease damages the feather, deforms the beak, and will eventually lead to death. (KATOH et al., 2010) The disease is caused by a small circovirus, which is a single-stranded DNA virus belonging to the Circoviridae family (Hakimuddin et al., 2016). The virus can spreads through direct contact with contaminated surfaces, feces, feather dander, and other bodily excretions (Greenacre, 2005). It can be transmitted horizontally to other birds in the same generation and vertically to eggs and young chicks in the next generation (Hakimuddin et al., 2016). Since the virus has a non-envelope structure, it is able to resist many control measures and is able to persist in the environment and infected substances for a long time. The origin of PBFD was thought of to be from Australia (PASS & PERRY, 1984), where it then spread to the rest of the world. Possibly through pet trades and import of these birds, this disease was able to spread globally. Report of this disease has occurred in other countries located in North America, Europe, Africa, Asia, and even on islands in the Indian and Pacific Oceans (Harkins et al., 2014). Psittacine beak and feather disease prevalence around the world varies and has been reported to be around 41.2% in Taiwan (Hsu et al., 2006), 3.5–4% in USA (de Kloet & de Kloet, 2004) and 23% in Australia (Khalesi et al., 2005). With an increasing trend of live birds being traded globally, the spread of PBFD and other diseases will surely grow. Diagnosis Typical clinical signs of PBFD include lethargy, weight loss, shedding and abnormal development of feathers, beak elongation and deformation, and eventually death (PASS & PERRY, 1984). This disease can occur in three different forms: peracute, acute, and chronic. Progression of the disease depends on the age, with younger birds having a higher progression rate (Greenacre, 2005). Some symptoms of peracute PBFD are weight loss, pneumonia, sepsis, enteritis, liver necrosis, and leukopenia (Schoemaker et al., 2000). Sudden death is likely to occur in peracute PBFD. In acute PBFD, majority of those affected by this phase are between the ages of 0-3 years old and it is thought that their susceptibility is based on their condition instead of the virus’ antigenic or genotypic characteristics (Ritchie et al., 1990). Some clinical signs includes depression and rapidly developing feather dystrophy can occur, affecting 80-100% of the feathers in as little as one week (Ritchie, 1995). Sudden death can also occur in this form. Those that survive this phase will have an incubation period, which may be years, before going to the chronic PBFD phase (Greenacre, 2005). For chronic PBFD, it is typically characterized by symmetrical feather dystrophy that progresses slowly and gets worse over time (Greenacre, 2005). Birds can become completely bald and can have beak deformities (Figure 1), where the beak becomes elongated. Death usually occurs from secondary infections, fungal or bacterial, because lymphoid tissues are usually damaged by the virus and causes the immune system to be suppressed (Ritchie et al., 2003). Figure 1: Cockatoo with advanced PBFD (Harcourt-Brown, 2009) PBFD can be diagnosed successfully from just careful examination. The disease can first be suspected if the bird is progressively losing feathers or has a symmetrical feather dysplasia. However, a loss of feathers does not always mean it is PBFD as the cause can be from other reasons, such as being self-inflicted or from excessive allopreening, which causes injuries that look similar to those caused by the disease (Wellehan et al., 2016). PBFD can be diagnosed though antigen and antibody detection from hemagglutination assay and hemagglutination inhibition. In addition, polymerase chain reaction (PCR) is also used for detecting PBFD, being a standard method of detection in most countries (Wellehan et al., 2016). False positives can occur with this method due to the nature of the virus to easily contaminate and persist in the environment, which will contaminate the samples, such as feathers, that are exposed to this environment (Wellehan et al., 2016). Therefore, the choice of sample collection method can have a major impact to the results. In one study, it was found that the use of blood samples for a PCR test resulted in 47 out 56 birds being positive for PBFD, while only 10 birds had a positive result when feather samples were used (Khalesi et al., 2005). Treatment and Disease Control Current treatment for PBFD is for supportive care to prevent secondary infections as there is no cure for this disease. The disease is fatal when clinical signs appear, while other birds that has an immune response and don’t show any clinical signs, making this naturally vaccinated (Greenacre, 2005). Effective methods of controlling this disease involves isolating suspected carriers, testing, and if necessary, culling to prevent a possible outbreak from occurring. The resilience of the virus to many chemical disinfectants and even extreme temperatures can be based on the physicochemical properties of the virus (Raidal & M.Cross, 1994). However, Virkson S or other peroxide disinfectants have been suggested for use to disinfect contaminated areas (Wellehan et al., 2016). Strict hygiene practices with the right disinfectants is the key to prevent further spread of PBFD. While there are no vaccine for PBFD available commercially, research into developing one is ongoing and currently made vaccines appears to be effective. Future control of the disease will still depend on implementing strict hygiene practices and testing methods since vaccinated birds may still spread the disease. References Greenacre, C.B. (2005) Viral diseases of companion birds. Veterinary Clinics of North America: Exotic Animal Practice, 8, 85–105. KATOH, H., OGAWA, H., OHYA, K. & FUKUSHI, H. (2010) A review of DNA viral infections in Psittacine birds. Journal of
Introduction to Feline Hypertrophic Cardiomyopathy
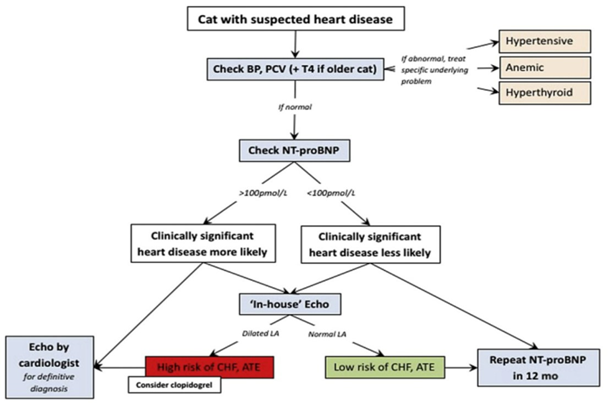
Maigan Espinili Maruquin It is important to be aware that some of the diseases your pets may have are actually inherited. In cats, there are myocardial diseases that can be breed- related. The most common myocardial disease in cats is Hypertrophic cardiomyopathy (HCM), wherein abnormal thickening of the walls of the left ventricle (LV) is observed [1]. First time described in cats in 1977 [2], it has been reported to have a prevalence of around 14.7% in apparently healthy cats [3-5]. In humans, the HCM is considered a genetic disease [6-8], whereas occurrences of the disease were observed in mix- breeds [9], Persian [10], and American shorthair cats [11], while an HCM caused by mutation was identified in Maine coon [12] and ragdoll [13]. The HCM are diagnosed at mean of 5-7 years, although all ages can get the disease [6]. On the other hand, some cat breeds including Maine Coons [14]; Sphynx [15], and Ragdoll [16] were reported on earlier onset of under 2 years old [3]. Cats that are diagnosed with HCM are also recorded to develop congestive heart failure (CHF), arterial thromboembolism (ATE), or sudden cardiac death (SCD) [1, 17, 18]. Clinical Presentation When cats visit the clinics, routine veterinary examinations are conducted, and during auscultation, signs like arrhythmias, gallop sounds, or murmurs can be detected [6, 19, 20]. Respiratory distress is a manifestation of heart failure in diseased cats, whereas, some cats display hypothermia and pre-renal azotemia. On the other hand, the murmurs in cats may vary in intensity form moment to moment, and are commonly associated with dynamic and labile phenomena [6]. Diagnosis Fig. 1. Approach to the asymptomatic cat with suspected heart disease. BP, blood pressure; PCV, packed cell volume; T4, thyroxine [1] The feline HCM are primarily diagnosed on echocardiographic examination, which recognizes basic patterns that are intuitive [21], with ventricular wall thickness that is equal to or exceed 6 mm [6, 22]. Respiratory distress is reported to display left atrial enlargement. However, echocardiographic examination has limitations [1] and there is no definitive, gold-standard to diagnose HCM, unless there is a hypothetical and flawless molecular or genetic testing [6]. The LV wall thickness has no exact value allowable, and body weight can affect its thickness [1]. An increase of cTn-I in plasma concentration indicates its sensitivity and specificity as a biomarker to provide myocardial damage severity and prognosis information. On the other hand, the N-terminal pro B-type natriuretic peptide (NT-proBNP) assay may provide ongoing myocardial stress, however, full cardiac evaluation shall be performed to detect its cause of elevation [1]. Myocyte enlargement and interstitial fibrosis were observed, along with disorganized spatial arrangement of myocytes in histopathological examination [3, 23] Genetic testing for single point mutation that affects MYBPC3 in Maine coon cats (A31P) [12] and ragdolls (R820W) [13] are commercially available. Autosomal dominant inheritance were reported in both breeds [1]. Therapy and Management For asymptomatic cats with HCM, diltiazem or beta-blockers were reported to improve physical condition. Meanwhile, Diltiazem is administered at three times a day as a licensed formulation in UK to manage cases of HCM [21]. In a study conducted by Rishniw, M. and P.D. Pion in 2011, participatiing clinicians used furosemide for evident CHF, and most of them also used and ACEIs, while for cases with substantial dynamic LVOT obstruction, β-blockers were used by most [24]. Altering the progression of HCM in the pre- or subclinical stage is an approach that is ideal in the absence of safe and efficient therapy [1]. References Luis Fuentes, V. and L.J. Wilkie, Asymptomatic Hypertrophic Cardiomyopathy: Diagnosis and Therapy. Veterinary Clinics: Small Animal Practice, 2017. 47(5): p. 1041-1054. Tilley, L.P., et al., Primary myocardial disease in the cat. A model for human cardiomyopathy. Am J Pathol, 1977. 86(3): p. 493-522. Gil-Ortuño, C., et al., Genetics of feline hypertrophic cardiomyopathy. 2020. 98(3): p. 203-214. Paige, C.F., et al., Prevalence of cardiomyopathy in apparently healthy cats. J Am Vet Med Assoc, 2009. 234(11): p. 1398-403. Payne, J.R., D.C. Brodbelt, and V. Luis Fuentes, Cardiomyopathy prevalence in 780 apparently healthy cats in rehoming centres (the CatScan study). J Vet Cardiol, 2015. 17 Suppl 1: p. S244-57. Abbott, J.A., Feline Hypertrophic Cardiomyopathy: An Update. Veterinary Clinics: Small Animal Practice, 2010. 40(4): p. 685-700. Maron, B.J., et al., American College of Cardiology/European Society of Cardiology clinical expert consensus document on hypertrophic cardiomyopathy. A report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus Documents and the European Society of Cardiology Committee for Practice Guidelines. J Am Coll Cardiol, 2003. 42(9): p. 1687-713. Maron, B.J., Hypertrophic cardiomyopathy: a systematic review. Jama, 2002. 287(10): p. 1308-20. Kraus, M.S., C.A. Calvert, and G.J. Jacobs, Hypertrophic cardiomyopathy in a litter of five mixed-breed cats. J Am Anim Hosp Assoc, 1999. 35(4): p. 293-6. Marin L, V.S., Boon J, et al., Left ventricular hypertrophy in a closed colony of Persian cats [abstract]. J Vet Intern Med 1994. 8:143. Meurs KM, K.M., Towbin J, et al., Familial systolic anterior motion of the mitral valve and/or hypertrophic cardiomyopathy is apparently inherited as an autosomal dominant trait in a family of American shorthair cats. J Vet Intern Med, 1997. 11:138. Meurs, K.M., et al., A cardiac myosin binding protein C mutation in the Maine Coon cat with familial hypertrophic cardiomyopathy. Hum Mol Genet, 2005. 14(23): p. 3587-93. Meurs, K.M., et al., A substitution mutation in the myosin binding protein C gene in ragdoll hypertrophic cardiomyopathy. Genomics, 2007. 90(2): p. 261-4. Kittleson, M.D., et al., Familial hypertrophic cardiomyopathy in maine coon cats: an animal model of human disease. Circulation, 1999. 99(24): p. 3172-80. Chetboul, V., et al., Prospective echocardiographic and tissue Doppler screening of a large Sphynx cat population: Reference ranges, heart disease prevalence and genetic aspects. Journal of veterinary cardiology : the official journal of the European Society of Veterinary Cardiology, 2012. 14. Borgeat, K., et al., The influence of clinical and genetic factors on left ventricular wall thickness in Ragdoll cats. J
Feline Hyperthyroidism

Sushant Sadotra Introduction: One of the most common diseases in middle-aged and older cats is hyperthyroidism. An increase in the production of thyroid hormones, i.e., T4 and T3, is the primary cause of this disorder. The enlarged thyroid gland in the neck region of the cat is the most common visible sign identifying hyperthyroidism. This enlargement is a non-cancerous tumor known as an adenoma. However, in some rare cases, it can also be caused by malignant tumors known as thyroid adenocarcinomas. Hyperthyroidism, also called thyrotoxicosis, increases the metabolic rate in an animal’s body because of high circulating thyroid hormone and often causes secondary problems by affecting all of the organs in the body. The reason for feline hyperthyroidism is unfamiliar. However, deficiencies or excesses of some elements in the diet and thyroid-disrupting could be responsible for the onset of hyperthyroidism. Hyperthyroidism is rare in dogs. However, if it occurs, it is primarily because of thyroid carcinoma. This contrasts with the case in hyperthyroid cats, where less than 5% of a thyroid tumor is carcinoma. Clinical signs: Cats’ most common clinical signs of hyperthyroidism are weight loss, increased appetite, vomiting, diarrhea, increased fecal volume, hyperexcitability, polydipsia, polyuria, enlargement of the thyroid gland, cardiomegaly, and congestive heart failure. Diagnosis A high thyroid hormone concentration in serum is the primary indication of hyperthyroidism. Therefore measuring serum total T4 concentration is the standard procedure that can confirm the diagnosis of hyperthyroidism in cats. In 5% – 10% of cases, it is also seen that cats so have normal T4 levels. It could be an indication of early or mild hyperthyroidism. Suppression of a high total T4 level to within reference range limits can also be caused by a nonthyroidal illness concurrent with hyperthyroidism. High free T4 concentration, medical history, and physical examination diagnose hyperthyroidism in cats with normal T4 levels. Treatment Radioiodine Therapy The radioiodine can be concentrated within the thyroid gland, where it targets the tumor by selectively irradiating and destroying hyper-functioning tissue. Radioactive iodine therapy is a simple, effective, and safe treatment for cat hyperthyroidism. Thyroidectomy Unilateral thyroid tumors can be easily treatable with surgical thyroidectomy without requiring thyroxine supplementation. Thyroidectomy can also be used for bilateral thyroid tumors. However, to avoid postoperative hypocalcemia, the functioning of the parathyroid gland must be preserved. After complete thyroidectomy, thyroxine should be administered for one to two days. Vitamin D and calcium treatment are also indicated if iatrogenic hypoparathyroidism develops. Chronic administration of an antithyroid drug An antithyroid drug acts by blocking thyroid hormone synthesis. Methimazole, carbimazole, and propylthiouracil are some of the most commonly used antithyroid drugs that are used to control hyperthyroidism. An initial dose of methimazole is 2.5 mg to 5 mg and is divided into two equal amounts to be given daily. Propylthiouracil has shown some adverse effects (hemolytic anemia and thrombocytopenia) and therefore is not recommended in cats. In less than 5% of treated cats, methimazole may have adverse effects such as agranulocytosis and thrombocytopenia. Besides lowering the circulating T4 concentration, cardiovascular signs such as tachycardia, tachypnea, hypertension, and hyperexcitability are often treated using β-adrenoceptor blocking agents such as propranolol and atenolol. Another strategic drug that inhibits the conversion of peripheral T4 to T3 is the oral cholecystographic agents such as ipodate, iopanoic acid, and diatrizoate meglumine. Lifelong nutritional therapy A diet with iodine levels below the minimum daily requirement is mainly prescribed for cats that are not suitable as a candidate for surgery or radioiodine therapy or in cats that develop adverse effects from oral medication. Compared to cats with severe hyperthyroidism, this nutritional therapy is more effective if the cat has a moderate increase in T4 levels. Hill’s® y/d Feline Thyroid HealthTM is one of the prescription diets available in the market with severely restricted iodine levels. The therapy can only control but not wholly treat hyperthyroidism. Also, cats on an iodine-deficient diet must not eat any other diet or food. If the diet is stopped, a setback will occur, and the therapy will be ineffective in regulating hyperthyroidism. References Vaske, Heather H et al. “Diagnosis and management of feline hyperthyroidism: current perspectives.” Veterinary medicine (Auckland, N.Z.) vol. 5 85-96. 20 Aug. 2014, doi:10.2147/VMRR.S39985 Carney, Hazel C et al. “2016 AAFP Guidelines for the Management of Feline Hyperthyroidism.” Journal of feline medicine and surgery vol. 18, 5 (2016): 400-16. doi:10.1177/1098612X16643252 Mark E. Peterson. Hyperthyroidism in Animals. Last full review/revision Jul 2019 | Content last modified Oct 2020. MSD MANUAL Veterinary Manual.
Canine Ehrlichiosis
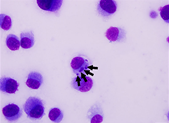
Oliver Organista, LA Ehrlichiosis (also known as canine rickettsiosis, canine hemorrhagic fever, canine typhus, tracker dog disease, and tropical canine pancytopenia) is a tick-borne disease of dogs usually caused by organism Ehrlichia canis. Ehrlichia canis was first identified in 1935 in Algeria; dogs infested with ticks showed fever and anemia [1]. Later, during the Vietnam War, many military working dogs brought to Vietnam by the US army exhibited a severe disease called Tropical Canine Pancytopenia [3]. Later, it was renamed canine monocytic ehrlichiosis (CME). Canine monocytotropic ehrlichiosis (CME), caused by the rickettsia E. canis, is an important canine disease with a worldwide distribution. Diagnosis of the disease can be challenging because of its different phases and multiple clinical manifestations. CME should be suspected when a compatible history (living in or traveling to an endemic region, previous tick exposure), typical clinical signs and characteristic hematological and biochemical abnormalities are present [4]. German shepherd dogs are thought to be particularly affected by the disease, other breeds generally have milder clinical signs. Cats can also be infected [2] Figure 1a. Peripheral blood smear from a dog. On the feathered edge, monocytes rarely contain cytoplasmic morula (arrow) consistent with Ehrlichia canis. Source: https://tvmdl.tamu.edu/2019/10/01/ehrlichia-canis-discovered-in-dog/. 1b. Photomicrograph of an E. canis strain isolated from the leukocytes of a dog Uberlandia, Brazil. Andia,dia, , Brazil. Note the multiple mourale in the DH82 cell cytopolasm (arrows). Source: DOI:10.4142/jvs.2014.15.2.241 Ehrlichiosis symptoms in dogs can be classified into three stages: early disease (acute phase), sub-clinical (no outward appearance of disease), and clinical or chronic (long-standing infection) [5]. Acute Phase The acute stage of ehrlichiosis can last 2 to 4 weeks, during which the infection is either eliminated or your dog will progress to the sub-clinical phase. Symptoms of the acute stage include: Swollen lymph nodes Weight loss Respiratory distress Bleeding disorders (spontaneous hemorrhage or bleeding) Fever Neurological disturbances (meningitis or unsteady on feet) Sub-Clinical Phase In the sub-clinical phase, the Ehrlichia organism is present but there may not be any outward signs of disease. This is often considered the worst phase of the disease because it is able to progress undetected. In some cases the disease is detected when the vet notices prolonged bleeding from the injection site after taking a blood sample. If the organisms are not eliminated in this stage, your pooch’s infection may move to the next stage – clinical ehrlichiosis. Clinical Phase Clinical ehrlichiosis happens when the organism isn’t eliminated by the immune system in one of the previous stages. This stage can lead to several serious symptoms, including: Lameness Swollen limbs Neurological problems Bleeding episodes Anemia Eye problems (such as blindness or hemorrhage into eyes) This phase becomes extremely serious if the bone marrow (where blood cells are produced) fails, meaning that your pup will be unable to create any blood cells he needs to sustain life (platelets, white blood cells and red blood cells). Diagnosis can be performed using visual, serologic, or molecular methods. Erhlichia spp. replicate inside a membrane-bound vacuole (i.e., morula) that can sometimes be observed by light microscopic examination of stained blood smears inside either monocytes (E. canis and E. chaffeensis) or granulocytes (E. ewingii). A cross-reactivity between antibodies is possible when performing an immunofluorescent assay (IFA) or enzyme linked immunosorbant assays (ELISA) [7]. The most common molecular method used to diagnose an Ehrlichia spp. infection, particularly in dogs with acute illness where the onset of clinical signs may precede a measurable antibody response is by polymerase chain reaction (PCR) [8]. In Canines, blood counts and hematological tests are crucial to the diagnosis of Canine monocytic ehrlichiosis. Low blood counts or thrombocytopenia are often critical signs of ehrlichial infection. In a study conducted by Shipov et. al red blood cell count and hemoglobin were used as the diagnostic indicators for canine ehrlichiosis (2008). Within the study dogs were assigned to two categories: survivors and non- survivors. Within each group the prognostic indicators and treatment protocols were observed and compared. Most commonly the dogs under examination exhibited symptoms such as weakness pale mucous membranes, fever, and bleeding tendencies [9]. Ehrlichiosis is typically treated with a 28- to 30-day course of antibiotics, most often prescribing doxycycline [11,14]. Doxycycline tablets were administered orally once a day for 20 consecutive days, at a target dose level of 10 mg/kg. The actual dose administered was calculated as ranging between 10 mg/kg and 11.7 mg/kg [12] .Most dogs in the acute or subclinical phases will not require hospitalization and can be managed as outpatients at home with minimal supportive care (pain medications and appetite stimulants). Dogs with chronic ehrlichiosis may require hospitalization for aggressive supportive care that includes blood transfusions, steroids, IV fluids, and nutritional support [10]. Minocycline can be an effective alternative to doxycycline for clearing E. canis from the blood in nonacute infections [13]. Fortunately, most tick bites can be prevented through monthly flea and tick preventative care. There are plenty of options available, including topical, tablet, and chewable medications. Your veterinarian will be able to help you find the best option for your pet. If you live near wooded areas where ticks are prone, it is best to keep your dog away from these areas, since there currently is no vaccine for ehrlichiosis. When your dog comes back from any outdoor adventure, it’s important to inspect them for any ticks or fleas and remove them safely. Early removal of ticks is the best defense against the spread of any infection [15]. Reference Donatien, A., and F. Lestoquard. “Existence en Algerie d’une Rickettsia du chien.” Bull. Soc. Pathol. Exot 28 (1935): 418-419. Eddlestone, S.M., et al., Doxycycline clearance of experimentally induced chronic Ehrlichia canis infection in dogs. J Vet Intern Med, 2007. 21(6): p. 1237-42. Huxsoll DL, Hildebrandt PK, Nims RM, Walker JS. Tropical canine pancytopenia. J Am Vet Med Assoc. 1970;157(11):1627–32. Harrus S, Waner T. Diagnosis of canine monocytotropic ehrlichiosis (Ehrlichia canis): an overview. Vet J. 2011 Mar;187(3):292-6. doi: 10.1016/j.tvjl.2010.02.001. Epub 2010 Mar 11. PMID: 20226700. Veterinary Specialty Center Tucson (www.vscot.com), Ehrlichiosis in
Feline Parvovirus (FPV) Guide: Symptoms, Diagnosis & Prevention
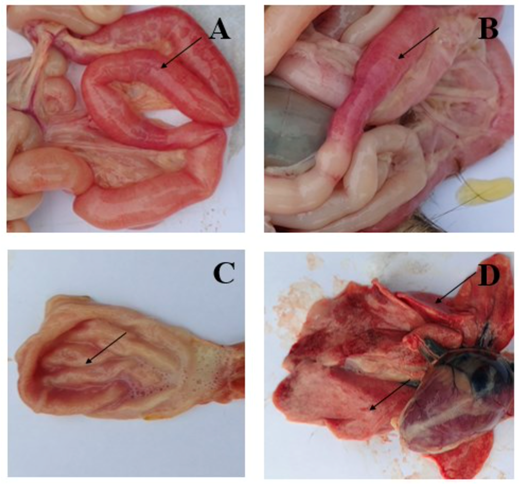
Table of Contents Trinh Mai Nguyen Tang 1. Introduction Feline panleukopenia (FP), commonly referred to as feline distemper, is a globally significant and often fatal infectious disease of cats caused by feline panleukopenia virus (FPV). Domestic cats are considered the primary host of the virus. Taxonomically, FPV is a non-enveloped, single-stranded DNA virus belonging to the family Parvoviridae, genus Protoparvovirus, and species Carnivore protoparvovirus 1. Infection with FPV results in feline leukopenia disease and is characterized by high contagiousness and substantial lethality in susceptible feline populations. The existence of FPV has been recognized for over a century. The disease was first described in cats in the early twentieth century and formally reported in France in 1928 by Verge and Christoforoni. Since that time, FPV has become endemic worldwide, with disease incidence often following a distinct seasonal pattern. Reported cases typically increase from early summer through autumn, with peak prevalence occurring during July, August, and September. A major challenge in FPV control lies in the virus’s exceptional environmental durability and resistance to inactivation. As a non-enveloped virus, FPV is highly resistant to many commonly used chemical disinfectants, including alcohols, iodine-based products, and quaternary ammonium compounds, despite frequent label claims of virucidal activity. FPV can persist in contaminated environments for months or even years, contributing to repeated exposure and reinfection risks. Effective environmental decontamination therefore requires the use of strongly oxidizing disinfectants, such as a 1:32 dilution of household bleach (sodium hypochlorite), potassium peroxymonosulfate (for example, Virkon at 1:100 dilution), or accelerated hydrogen peroxide formulations. From a clinical perspective, FPV remains a major concern due to its high transmissibility and elevated mortality rates, particularly in unvaccinated cats and young kittens. The virus exhibits a marked tropism for rapidly dividing cells, including those in the bone marrow, lymphoid tissues, and intestinal crypt epithelium. This selective cellular targeting leads to profound panleukopenia, severe gastroenteritis characterized by vomiting, dehydration, and, in some cases, hemorrhagic diarrhea, as well as significant immunosuppression. In kittens and inadequately vaccinated cats, mortality rates frequently exceed 90%. The combination of extreme environmental persistence, high infectivity, and severe clinical outcomes makes FPV a persistent and serious threat, particularly in high-density settings such as animal shelters, where outbreaks can result in widespread morbidity, mortality, and substantial operational challenges. 2. Transmission and Clinical Signs of FPV 2.1 Transmission Pathways and Cellular Entry Feline panleukopenia virus (FPV) is transmitted primarily via the fecal–oral route. Infection occurs when susceptible cats ingest viral particles through contaminated food, water, fomites, or contact with infected secretions. Because of the virus’s remarkable environmental stability, indirect transmission via contaminated cages, litter boxes, bedding, transport carriers, and human hands or clothing plays a major role in disease spread, particularly in high-density environments. At the cellular level, FPV initiates infection by binding to the feline transferrin receptor (fTfR) on the host cell surface, which facilitates viral entry and internalization [6–7]. This receptor-mediated process largely determines host specificity and tissue tropism and explains the virus’s predilection for rapidly dividing feline cells. 2.2 Pathogenesis and Tissue Tropism FPV exhibits a strong tropism for rapidly proliferating cells, a hallmark feature of parvoviral infections. Previous studies have demonstrated that the virus replicates predominantly in small intestinal crypt epithelial cells and lymphoid cells [8–9]. Viral replication within these tissues disrupts normal cell turnover and immune function. In the gastrointestinal tract, destruction of crypt epithelial cells leads to villous collapse, impaired absorption, and breakdown of the intestinal barrier. Concurrently, infection of lymphoid tissues and bone marrow results in severe leukopenia, compromising innate and adaptive immune responses. Hemorrhage within the small intestine and stomach contributes to the development of hemorrhagic diarrhea, a characteristic and often life-threatening manifestation of FPV infection [10]. 2.3 Gross Lesions and Organ Involvement Gross pathological findings in FPV-infected cats reflect the systemic nature of the disease. In a pathological study reported by Yen et al. (2021), examination of eight FPV-infected cats revealed small intestinal congestion in 100% of cases, with intestinal mucosal ulceration observed in 75% of the animals (Figure 1A–B). Gastric congestion was present in 75% of cases (Figure 1C), while mild pneumonia was detected in 25% of cats (Figure 1D). Additional lesions reported included enlarged mesenteric lymph nodes and splenic infarction, consistent with widespread lymphoid depletion and vascular compromise [10]. These findings underscore the multisystemic involvement of FPV and help explain the rapid clinical deterioration observed in severe cases. Figure 1. Symptoms of FPV infection in cats. A, B: Intestinal congestion with scattered or intermittent bleeding. C: Congested stomach containing large volumes of fluid. D: Mild pneumonia. 2.4 Clinical Progression and Systemic Effects The pathological changes induced by FPV give rise to a characteristic progression of clinical signs. Early manifestations often include anorexia, lethargy, fever, vomiting, and deterioration of coat condition. As the disease advances, affected cats frequently develop profuse diarrhea, severe dehydration, electrolyte imbalances, and hypoglycemia. In advanced stages, disruption of the intestinal barrier and profound immunosuppression predispose cats to bacterial translocation, sepsis, and endotoxemia. Without timely and intensive supportive treatment, these systemic complications can lead to rapid clinical decline and death [12–15]. The severity and speed of disease progression are particularly pronounced in kittens and immunologically naïve cats. 2.5 Neurological Manifestations in Kittens In addition to gastrointestinal and hematological effects, FPV infection can result in neurological abnormalities, particularly when infection occurs in utero or during early neonatal development. Reported central nervous system lesions include cerebellar hypoplasia and hydrocephalus, which arise from viral interference with neuronal precursor cell division. Kittens affected by cerebellar hypoplasia may survive the acute infection but exhibit permanent neurological deficits, such as ataxia, intention tremors, and impaired coordination. These manifestations are non-progressive but irreversible and represent an important long-term consequence of early FPV exposure. 2.6 Incubation Period and Viral Shedding The incubation period of FPV infection is typically 4 to 6 days, although clinical onset may vary depending on viral dose, host immunity, and age. During active infection, cats can shed large quantities of virus in feces for up to 43 days, contributing significantly to
Feline Polycystic Kidney Disease (PKD)
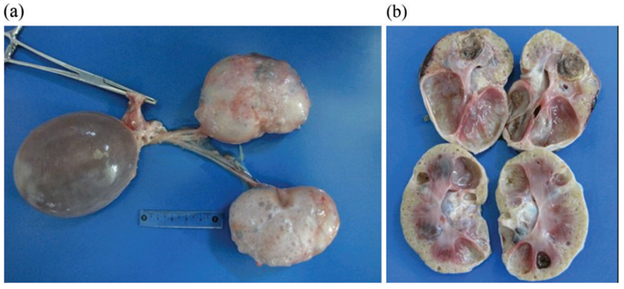
Maigan Espinili Maruquin Introduction Cats, as a fur family, require health attention. However, some felines can get infected with certain types of inherited diseases. One of the most prevalent genetic diseases is the Feline Polycystic Kidney Disease (PKD) [1, 2], which causes the progressive development of multiple fluid-filled cysts in the kidney and in some cases extends to the liver and pancreas [2-5]. Prevalence of the PKD in Persian cats has been studied in different countries including, the United Kingdom (49.2%) [6], Japan (46%) [7], Australia (45%) [8], and France (40.45%) [9]. Reports suggest that the disease is not related to the sex of the feline, after results show no statistical differences between males and females [9, 10]. Different species of felines can get the said disease like Charteux [11], Neva Masquerade cat [12], and Scottish Fold and American Shorthair [7], however, it is most commonly inherited by the Persian and Persian- related breeds [3] with 35 to 57% detection via ultrasonography or genetic testing [13]. It is suspected that all the feline breeds could have inherited the mutation where the disease starts, considering that around 80% of all current feline breeds have had some type of cross with Persian breed [3, 14]. In humans, 1 in 500–1000 of the general population can inherit Autosomal dominant polycystic kidney disease (ADPKD) [15], which is characterized by increase in kidney size caused by the development and progressive enlargement of renal cysts, eventually leading to a steady decline in glomerular filtration rate [16]. The PKD1 and PKD2 genes, which are responsible for coding the polycystin-1 and 2 proteins, where 85 % and 15%, respectively, are responsible for the mutations [17]. Relative to the cases recorded with ADPKD in cats, the feline ortholog of the human PKD1 gene, which is also named PKD1, were recorded to have a single germline mutation. During the mutation, at exon 29 of the feline PKD1 gene, position 3284, a pyrimidine base (cytosine, C) is substituted by a purine base (adenine, A), wherein the premature stop codon produced causes the 25% loss in the C- Terminal formation of polycystin-1 protein [3, 18, 19]. With similar features clinically and morphologically in both human and feline ADPKD [2], feline PKD is a good representation for human ADPKD [7]. Clinical Presentations Fig. 1. Postmortem images, including cross-section of the polycystic kidneys of a seven-year-old, entire male British shorthair cat (cat 4), tested positive for the PKD1 mutation, demonstrating multiple cysts of variable sizes in both kidneys (a, b). [4] The polycystin-1 is suggested to play a role in cell–cell and matrix–cell interactions [20]. The primary cilium, where this protein is expressed, functions in fluid transport and chemo and mechanoreceptors [21, 22]. Cysts of different sizes were presented in renal cortex and medulla, sometimes occurs in the liver and pancreas, as well [8], and increases both in size and quantity as they age [23]. With slow growth and progression, affected cats start to deteriorate renal functions [7]. The cysts develop by different scenarios including the increase of cell proliferation, fluid secretion, and extracellular matrix alterations, cilia with lost polarization would then alter the water reabsorption function [3, 24]. Secondary to renal cysts development, obstruction due to nephrolithiasis, lymphoma and chronic kidney disease with interstitial nephritis are also displayed, especially in old cats [8, 25] In a study conducted among cats in Turkey, some diagnosed cats were reported for fatigue, anorexia, and vomiting. During palpation, an increase in total kidney volume was discovered, and cystic lesions were seen in the cortex of both kidneys when ultrasonography was performed [19]. Generally, apathy, anorexia, weight loss, bad appearance of the coat, polyuria and polydipsia, and gastrointestinal disorders could be observed [26-28], while general dehydration and pale mucous membranes are also noticeable on clinical examinations [3]. Elevated serum Cre concentration has been presented as one of the clinical signs in cats from three years old, while other cats at nine showed normal concentration. This suggests variable clinical courses of the disease [7]. Whereas, few cases showed hepatic cysts an extrarenal manifestation [6]. Diagnosis Imaging Diagnosis Fig. 2. Ultrasound picture of a 2.5 year old Persian cat, positive for PKD. Note the distal enhancement beyond the anechoic cyst structures (arrow). The largest cyst measures 29 mm in diameter [8]. The Ultrasound is known to be with the highest successful diagnosis, allowing quick and reliable diagnosis [23], and is considered due to availability and non-invasive, safe, cheap and effectivity in detecting the presence of kidney cysts [28]. Whereas, radiography and intravenous urography are usually used in more advanced cases, like when there is a presence of multiple, large cysts [3]. Molecular Approach Various PCR methods have been used to identify and amplify the DNA fragment of interest [3]. The RFLP-PCR was developed and used [18], real-time PCR or quantitative PCR is known to be reliable and faster than the earlier technique [29]. Whereas, ARMS-PCR (Amplification-refractory mutation system-Polymerase Chain Reaction) presented its advantages in time, low quantity of samples needed and its low cost, which has resulted in 100% sensitivity and specificity [30]. The synergistic use of genetic testing to confirm the presence of the causal mutation and make an early diagnosis; and the ultrasound to diagnose polycystic kidney disease and to monitor the progression of the disease has been agreed by several authors to plan detection programs for feline PKD [3, 30-32]. For as early as three years old, affected felines can develop signs of impaired renal function, and thus, positive cats are strongly discouraged for breeding [7]. References Young, A.E., et al., Feline polycystic kidney disease is linked to the PKD1 region. Mamm Genome, 2005. 16(1): p. 59-65. Eaton, K.A., et al., Autosomal dominant polycystic kidney disease in Persian and Persian-cross cats. Vet Pathol, 1997. 34(2): p. 117-26. Schirrer, L., P.J. Marín-García, and L. Llobat, Feline Polycystic Kidney Disease: An Update. 2021. 8(11): p. 269. Nivy, R., et al., Polycystic kidney
