Feline Polycystic Kidney Disease (PKD)
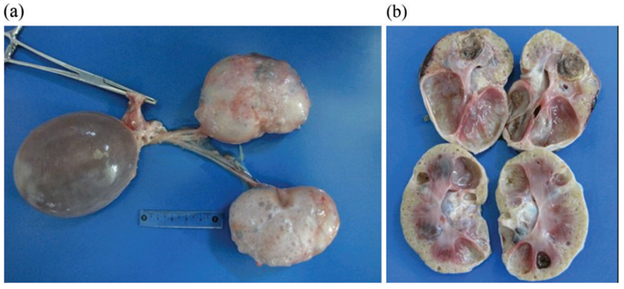
Maigan Espinili Maruquin Introduction Cats, as a fur family, require health attention. However, some felines can get infected with certain types of inherited diseases. One of the most prevalent genetic diseases is the Feline Polycystic Kidney Disease (PKD) [1, 2], which causes the progressive development of multiple fluid-filled cysts in the kidney and in some cases extends to the liver and pancreas [2-5]. Prevalence of the PKD in Persian cats has been studied in different countries including, the United Kingdom (49.2%) [6], Japan (46%) [7], Australia (45%) [8], and France (40.45%) [9]. Reports suggest that the disease is not related to the sex of the feline, after results show no statistical differences between males and females [9, 10]. Different species of felines can get the said disease like Charteux [11], Neva Masquerade cat [12], and Scottish Fold and American Shorthair [7], however, it is most commonly inherited by the Persian and Persian- related breeds [3] with 35 to 57% detection via ultrasonography or genetic testing [13]. It is suspected that all the feline breeds could have inherited the mutation where the disease starts, considering that around 80% of all current feline breeds have had some type of cross with Persian breed [3, 14]. In humans, 1 in 500–1000 of the general population can inherit Autosomal dominant polycystic kidney disease (ADPKD) [15], which is characterized by increase in kidney size caused by the development and progressive enlargement of renal cysts, eventually leading to a steady decline in glomerular filtration rate [16]. The PKD1 and PKD2 genes, which are responsible for coding the polycystin-1 and 2 proteins, where 85 % and 15%, respectively, are responsible for the mutations [17]. Relative to the cases recorded with ADPKD in cats, the feline ortholog of the human PKD1 gene, which is also named PKD1, were recorded to have a single germline mutation. During the mutation, at exon 29 of the feline PKD1 gene, position 3284, a pyrimidine base (cytosine, C) is substituted by a purine base (adenine, A), wherein the premature stop codon produced causes the 25% loss in the C- Terminal formation of polycystin-1 protein [3, 18, 19]. With similar features clinically and morphologically in both human and feline ADPKD [2], feline PKD is a good representation for human ADPKD [7]. Clinical Presentations Fig. 1. Postmortem images, including cross-section of the polycystic kidneys of a seven-year-old, entire male British shorthair cat (cat 4), tested positive for the PKD1 mutation, demonstrating multiple cysts of variable sizes in both kidneys (a, b). [4] The polycystin-1 is suggested to play a role in cell–cell and matrix–cell interactions [20]. The primary cilium, where this protein is expressed, functions in fluid transport and chemo and mechanoreceptors [21, 22]. Cysts of different sizes were presented in renal cortex and medulla, sometimes occurs in the liver and pancreas, as well [8], and increases both in size and quantity as they age [23]. With slow growth and progression, affected cats start to deteriorate renal functions [7]. The cysts develop by different scenarios including the increase of cell proliferation, fluid secretion, and extracellular matrix alterations, cilia with lost polarization would then alter the water reabsorption function [3, 24]. Secondary to renal cysts development, obstruction due to nephrolithiasis, lymphoma and chronic kidney disease with interstitial nephritis are also displayed, especially in old cats [8, 25] In a study conducted among cats in Turkey, some diagnosed cats were reported for fatigue, anorexia, and vomiting. During palpation, an increase in total kidney volume was discovered, and cystic lesions were seen in the cortex of both kidneys when ultrasonography was performed [19]. Generally, apathy, anorexia, weight loss, bad appearance of the coat, polyuria and polydipsia, and gastrointestinal disorders could be observed [26-28], while general dehydration and pale mucous membranes are also noticeable on clinical examinations [3]. Elevated serum Cre concentration has been presented as one of the clinical signs in cats from three years old, while other cats at nine showed normal concentration. This suggests variable clinical courses of the disease [7]. Whereas, few cases showed hepatic cysts an extrarenal manifestation [6]. Diagnosis Imaging Diagnosis Fig. 2. Ultrasound picture of a 2.5 year old Persian cat, positive for PKD. Note the distal enhancement beyond the anechoic cyst structures (arrow). The largest cyst measures 29 mm in diameter [8]. The Ultrasound is known to be with the highest successful diagnosis, allowing quick and reliable diagnosis [23], and is considered due to availability and non-invasive, safe, cheap and effectivity in detecting the presence of kidney cysts [28]. Whereas, radiography and intravenous urography are usually used in more advanced cases, like when there is a presence of multiple, large cysts [3]. Molecular Approach Various PCR methods have been used to identify and amplify the DNA fragment of interest [3]. The RFLP-PCR was developed and used [18], real-time PCR or quantitative PCR is known to be reliable and faster than the earlier technique [29]. Whereas, ARMS-PCR (Amplification-refractory mutation system-Polymerase Chain Reaction) presented its advantages in time, low quantity of samples needed and its low cost, which has resulted in 100% sensitivity and specificity [30]. The synergistic use of genetic testing to confirm the presence of the causal mutation and make an early diagnosis; and the ultrasound to diagnose polycystic kidney disease and to monitor the progression of the disease has been agreed by several authors to plan detection programs for feline PKD [3, 30-32]. For as early as three years old, affected felines can develop signs of impaired renal function, and thus, positive cats are strongly discouraged for breeding [7]. References Young, A.E., et al., Feline polycystic kidney disease is linked to the PKD1 region. Mamm Genome, 2005. 16(1): p. 59-65. Eaton, K.A., et al., Autosomal dominant polycystic kidney disease in Persian and Persian-cross cats. Vet Pathol, 1997. 34(2): p. 117-26. Schirrer, L., P.J. Marín-García, and L. Llobat, Feline Polycystic Kidney Disease: An Update. 2021. 8(11): p. 269. Nivy, R., et al., Polycystic kidney
Canine Hypothyroidism
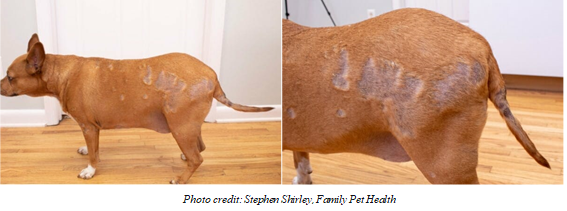
Sushant Sadotra Introduction: Hypothyroidism is a prevalent thyroid disorder that is caused due to the deficiency of thyroid hormone. In this condition, there is an irregular short production and improper secretion of thyroid hormones into the blood from the thyroid gland. This leads to a slow metabolic rate and loss of proper body functions. Regarding pet animals, hypothyroidism is mostly occurring among dogs and rarely in cats and other pet animals. Hypothyroidism Etiology in canines: Imbalance at any level of the hypothalamic-pituitary-thyroid axis can cause hypothyroidism in animals, especially dogs. In Adult dogs, the onset of primary hypothyroidism can cause either because of lymphocytic thyroiditis or idiopathic atrophy of the thyroid gland. The gradual destruction of follicles and secondary fibrosis on the gland because of diffuse infiltration by lymphocytes, plasma cells, and macrophages into the thyroid gland. This condition is known as lymphocytic thyroiditis. Loss of thyroid parenchyma leads to the idiopathic atrophy of the thyroid gland, in which fatty tissue will replace the lost thyroid parenchyma. The condition can occur due to autoimmune thyroiditis. Secondary hypothyroidism can be caused due to the damage of pituitary thyrotrophs by tumor growth, leading to the deficiency of one or more pituitary hormones. Hypothyroidism in dogs can occur due to other rare causes such as congenital hypothyroidism or neoplastic destruction of thyroid tissue. Congenital hypothyroidism can be primary or secondary. Thyroid dysgenesis and dyshormonogenesis can cause congenital primary hypothyroidism in dogs. Congenital secondary hypothyroidism can show clinical signs caused by the deficiency of growth hormones such as dwarfism, lethargy, gait abnormalities, or pituitary dwarfism. Diagnosis of hypothyroidism in canines: A variety of nonthyroidal factors and other conditions can mimic thyroid disorder and mislead the correct diagnosis of canine hypothyroidism. The severity and chronicity of the clinical findings associated with hypothyroidism and other clinicopathologic abnormalities of the hypothyroid state can be a basis for the choice of a proper diagnostic test. Tests that could confirm the diagnosis of hypothyroidism in canines are mentioned below: Total T4: The initial screening test for hypothyroidism can determine Total T4 concentration. A dog with a T4 concentration lower than the reference range limits may be anticipated to have a hypothyroid issue. However, it can also indicate that the dog can have a nonthyroidal illness such as sick euthyroid syndrome. Therefore, the total T4 test alone cannot be a proper diagnosis. Free T4: In serum, unbound T4 is supposed to be biologically active. Therefore, it is a very used test to differentiate between hypothyroid and euthyroid dogs. Also, a free T4 assay can give a better diagnosis with high sensitivity and specificity. Most commercial Free T4 tests use the method of single-stage solid phase (analogue) assays. However, an equilibrium dialysis step can improve the accuracy. Free T3: Among T4 and T3, T3 is the most potent hormone in animals. Therefore, measuring Free T3 is also a sensitive diagnostic step. However, during the onset of the primary hypothyroidism, serum T3 determination is weak. Also, it is found that in hypothyroidism dogs, serum T3 concentrations could be low, normal, or high. If the serum concentration of T3 is high, it is to be checked whether or not anti-T3 antibodies are producing false results in the T3 radioimmunoassay. Serum TSH concertation: In primary hypothyroidism, high serum TSH concentrations are expected that further may lead to low serum concentrations of T4 and free T4. A species-specific TSH assay can be used to check the high level of serum TSH. However, a false-negative result showing normal TSH concentrations may indicate a condition of primary hypothyroidism; a false-positive result showing high serum TSH concentrations may indicate a nonthyroidal illness in a euthyroid dog. A normal serum TSH concentration can indicate secondary hypothyroidism in a few cases. Therefore, serum TSH concentration results should always be coupled with other tests for definite confirmations. TSH stimulation test: In this particular test, bovine TSH is introduced exogenously into the dog’s body, and the thyroid gland’s response is evaluated. Firstly, a basal T4 is measured. Then bovine TSH is administrated at a dosage of 0.1 U/kg. After 6 hours, T4 levels are calculated for the second time to check the response of the thyroid gland. Result interpretations could be a no response for hypothyroidism, a normal and blunted response for the sick euthyroid syndrome. Although the TSH stimulation test is one of the accurate tests to check thyroid function, it is expensive and less available. Imaging: Thyroid Ultrasonography can detect the decreased echogenicity followed by decreased thyroid volume. The procedure can take the best imaging of the thyroid gland by technetium 99m (99mTc). This diagnostic tool can differentiate between hypothyroidism and euthyroid sickness. Therapeutic trial: In this approach, a thyroxine supplementation is given to a dog at a particular dosage. If the response is positive, the supplementation is stopped to check for the return of clinical signs related to hypothyroidism. This can confirm that the dog has thyroid-responsive diseases rather than other nonthyroidal issues. However, before starting a therapeutic trial, every attempt should eliminate nonthyroidal sickness. Also, therapeutic monitoring should be performed in case the therapy is unsuccessful. Treatment of hypothyroidism in canines: The typical treatment of hypothyroidism in dogs is the oral medication of levothyroxine (L-T4). L-T4 is a synthetic thyroid hormone that can restore blood thyroid hormone concentrations and reverse hypothyroidism’s effects. The replacement of natural with synthetic hormones will be used for the rest of the animal’s life. However, a great precaution is needed with the initial dose and tailoring of the drug. The dosages of L-T4 in d are 0.01–0.02 mg/lb (0.02–0.04 mg/kg). The drug is given every day once or twice without food. Reference: Hypothyroidism is the Most Common Hormone Imbalance of Dogs; Wendy Brooks, DVM, DABVP. Date Published: 02/26/2002, Date Reviewed/Revised: 08/16/2019. Strey S, Mischke R, Rieder J. Hypothyreose beim Hund: eine Übersicht [Hypothyroidism in dogs: an overview]. Tierarztl Prax Ausg K Kleintiere Heimtiere. 2021 Jun; 49(3):195-205. German. doi: 10.1055/a-1367-3387. Epub 2021 Jun 22. PMID: 34157761 Mark E. Peterson. Hypothyroidism in Animals. Last
Canine Distemper Virus
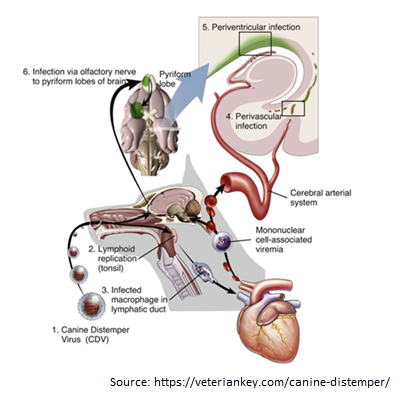
Canine Distemper Virus Tang Nguyen Mai Trinh Infection of canine distemper virus (CDV) is common in most terrestrial carnivores in the wild, especially in the Canidae family (eg, wolves, foxes, domestic dogs, etc.), as well as certain ferrets, otters, raccoons, cats, and even marine animals [1]. CDV is a member of the Paramyxoviridae family which the genus Morbilivirus was first described in the 1970s, however, it was only isolated in 1905 from the nasal secretions of infected dogs by a French veterinarian, [2]. CDV is now known as the most dangerous disease in dogs because of its propensity to infect through respiratory and digestive pathways and cause death in virtually all canines. When infected with it, the unvaccinated host has a 100% mortality rate [3]. Structure of Canine distemper virus CDV is known as a single-stranded RNA virus, like many other viruses CDV has an N nucleocapsid protein that is responsible for genome replication, matrix (M), fusion (F), phosphoprotein (P), and polymerase (L) which are involved in genomic and mRNA transcription. Whereas nonstructural proteins C and V have been identified as virulence factors involved in the modulation of immune responses and innate immunity, proteins like H and F are essential for viral attachment, invasion, and fusion into host cells [4-6]. Transmission Puppies and dogs most often become infected through airborne exposure to the virus from an infected dog or wild animal. CDV enter the dog’s body through respiratory secretions, most notably coughing and sneezing. Virus particles can spread about 25 feet with a single sneeze, then proliferate in immune cells such as macrophages, T and B cells via the SLAM receptor within 24 hours [8] and spreads to tonsils and lymph nodes via the lymphatic vessels, causing long-term immunosuppression [9-11]. The amount of virus in the lymph nodes of the throat, tonsils, and bronchi will go up after 2 to 4 days of infection, and the virus continues to release and replicate in multiple organs. This process takes 4 to 6 days after infection and affects organs such as the thymus, spleen, lymph nodes, stomach cells, lungs, and bone marrow. The increased concentration of virus in the lymph node tissue at this time causes the number of white blood cells to decrease, resulting in the infected dog’s body temperature increasing [12]. The virus begins to migrate to specific epithelium and the central nervous system (CNS) via the blood or CSF after around 8 –10 days [10]. Simultaneously, organs such as the respiratory tract, intestines, and urinary tract begin to experience significant infections as lymphocytes are destroyed, resulting in a massive release of viruses into these organs [13]. Figure 2. The process of entry and replication of CDV in dogs.1, The CDV virus enters the dog’s body through the respiratory tract. 2, the virus begins to multiply in the tonsils, bronchial lymph nodes, oropharyngeal nodes, and gastrointestinal lymphoid tissues. 3, the virus attacks immune cells such as macrophages and is released to other organs such as the heart. 4, The virus enters the central nervous system (CNS) through the circulatory system. 5, Virus enters the cerebrospinal fluid (CFS). 6, Viral migration to the pyramidal lobes of the cerebral cortex through the nasal passages to the central nervous system [14]. Clinical Symptoms of Canine Distemper Virus infection in dogs The CDV virus can cause disease in dogs of any age, although it is more common in pups aged 12 to 16 weeks and in unvaccinated dogs. Because pups’ immune systems are so immature, they are easily infected by the virus if they come into touch with dogs that have already been afflicted with CDV. When the dog get infected with the CDV virus, early symptoms include fever, weariness, lack of appetite, apathy, and a reluctance to be active [9]. However, these symptoms are quite similar to those of the parvovirus, which primarily infects the digestive system, whereas CDV not only assaults the digestive system but also damage to the respiratory system, neurological system, bones, arthritis, eyes, and skin [13]. Prolonged vomiting, diarrhea, severe dehydration, and stool abnormalities in color, consistency, and blood and mucus contribute to gastritis and intestinal inflammation in the early stages. Finally, the dog was unable to defecate, resulting in death (Figure 3A). Pus discharge, ulcers, and progressively becoming clouded eyes can lead to conjunctivitis and even blindness, while the skin appears as spots with pus within, then gradually changes from red to yellow (Figures 3 B and C). Signs such as a runny nose with green mucus in the early stage, shortness of breath in the latter stage, and shortness of breath lead to pneumonia in the respiratory system (Figure 3B). Figure 3. Signs of CDV infection in dogs. A, Dogs have digestive system damage, diarrhea with blood [17]. B, Eyes are damaged causing pus discharge, ulcers, while the nose shows signs of runny nose with green mucus [15]. C, The skin also appears spots that have pus inside. D and E, the dog’s paw and nose epithelium show basal keratinocytes. F, enamel deterioration in adult dogs following neonatal CDV infection [13]. When the disease is severe, evidence of central nervous system damage such as convulsions, hemiplegia, or quadriplegia will occur; at this stage, 99% of infected dogs will die. Furthermore, the CDV virus can attach to lymphocytes and enter the cerebrospinal fluid (CSF), causing harm to the central nervous system. In addition, CDV was discovered in the foot and nasal epithelium, promoting basal keratinocyte growth, which was clearly recognized in the Figures 3D and E. Although CDV infection leads to the above clinical signs, however the appearance of these signs is also dependent on habitat, age, host immunity, and virulence of the virus strain [13]. Studies have also demonstrated that CDV can be passed from mother to puppy through the placenta during pregnancy. When the mother dog is infected with the virus, it can lead to miscarriage, stillbirth, but this depends on the pregnancy stage of the mother dog [16]. For the lucky puppies
Canine Babesiosis
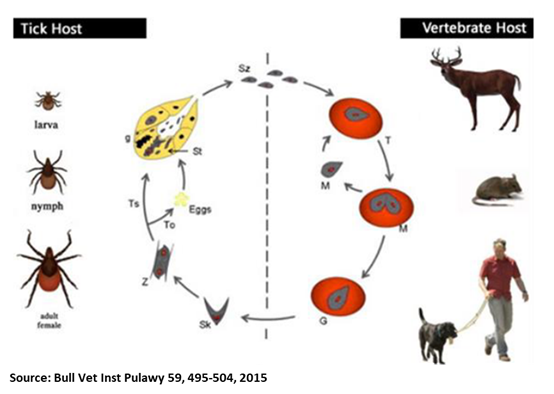
Oliver Organista, LA Canine babesiosis is a tick-borne, protozoal, haemoparasitic disease that can cause varying degrees of haemolytic anaemia, splenomegaly, thrombocytopenia and fever. It is very endemic to different parts of the world, and presents varying clinical, hematological, and pathological manifestation depending on the species and subspecies involved.1,2 There are two hosts for the transmission of Babesia spp., viz. invertebrate (tick) and vertebrate host. Dogs are one among the many targets of Babesia spp., causing canine babesiosis, and now there are clinical evidences of possible vertical transmission too. Dogs of all ages can be affected with Babesia spp., but young puppies are more commonly affected. Two Babesia parasites were thought to occur in dogs; the relatively large intra-erythrocytic piroplasm referred to as Babesia canis and a smaller parasite, known predominantly as the cause of canine babesiosis in Asia, Babesia gibsoni. B. canis has been reclassified into three separate species (B. canis, B. rosi, B. vogeli) on the basis of immunity, serological testing, vector specificity and molecular phylogeny At least 4 genetically and clinically distinct small piroplasms affect dogs which include: Babesia gibsoni–originally described in India nearly a century ago and now occurring sporadically in other parts of the world including the Australia; Babesia conradae, a piroplasm that occasionally infects dogs in California; Theileria annae, a Babesia microti-like parasite that has so far been reported only in northwest Spain, transmitted by Ixodes hexagonus ; and a fourth small piroplasm, B. (=T.) equi has also been reported in dogs in Spain.³ Prevalence of Babesiosis The incidence of canine babesiosis vary considerably from one country to another depending on the distribution of the causative parasite and their specific vectors. Table below shows the data gathered during the Questionnaire-based survey on the distribution and incidence of canine babesiosis in countries of Western Europe.5 DIAGNOSIS, SYMPTOMS, AND TREATMENT Diagnosis. In the past, babesiosis was diagnosed by seeing the parasite on a blood smear. Other diagnostic tests are becoming more readily available, including FA (fluorescent antibody) staining of the organism and ELISA (enzyme-linked immunosorbent assay) tests. A PCR (polymerase chain reaction) test is also available and is commonly used to diagnose babesiosis. The PCR test has the advantage in that it can detect all four species of Babesia. Serologic or antibody titer testing in the diagnosis of babesiosis has limitations. A positive test result is dependent on an antibody response by the infected dog, which may take up to ten days to develop. Once a dog has developed antibodies to babesiosis, they may persist for years and this must be considered when performing follow-up tests.6 Symptoms. Dogs typically present with the acute, severe form of babesiosis, which is characterized by findings such as abnormally dark urine, fever, weakness, pale mucous membranes, depression, swollen lymph nodes, and an enlarged spleen. Potential complications: Bleeding problems, a severe type of anemia that leads to jaundice, organ damage, death.7 Treatment. The FDA approved treatment for babesiosis is imidocarb diproprionate. A combination therapy of quinine, azithromycin, atovaquone, and/or clindamycin is being researched and may become more common to treat dogs within the US or Canada in the future. Clindamycin, the treatment of choice for Babesia microti, the main Babesia species that infects humans, can also be used against Babesia in dogs. Clindamycin is a readily available antibiotic and is an excellent starting point for treatment in many dogs. Your veterinarian will discuss any alternative and adjunctive treatments with you.7 TRANSMISSION AND PREVENTION OF BABESIOSIS How is dog Babesia transmitted? Transmission. In most cases, Babesia organisms are spread to dogs through the bite of an infected tick but is not likely to be transmitted until a tick is attached for 36 hours. However, some studies suggest that infected dogs with open mouth sores can pass on the infection to other dogs through a bite, and infected pregnant females can transmit babesiosis to their unborn puppies. It can also be transmitted by the needle passage of infected blood, inadvertently in the case of blood transfusion or deliberately during experimental studies. Generic Life Cycle of Babesia spp.10 Generic life cycle of Babesia spp. Sporozoites (Sz) are injected into a vertebrate host blood system, during the blood meal of an infected tick. After invasion, Sz differentiate into trophozoites (T). Trophozoites undergo asexual division into two or four merozoites (M) in the infected red blood cells. Merozoites exit the red blood cells and invade new ones. Some groups of merozoites transform into gamonts or pregamotyces (G). The process of gamogony and sporogony takes place in the tick. Gamonts ingested by a tick feeding on an infected host differentiate in the gut into gametes (called ray bodies or Strahlenkorper – (Sk)) that fuse forming a diploid zygote (Z, gamogony). Via meiosis division, zygotes give rise to motile haploid kinetes. After haploid kinetes multiply by sporogony, they penetrate the tick haemolymph and organs. The final stage of the development occurs in the salivary glands (Sg), where differentiation and multiplication occur. Kinetes transform into sporozoites that infect the vertebrate host after vector development into a subsequent life stage – larvae to nymph, nymph to adult (transstadial transmission, Ts). In large Babesia spp. kinetes also invade the tick ovaries and eggs, and infective sporozoites are formed in the salivary glands of the next generation larvae. This process is called transovarial transmission (To). Adopted and reproducted from Schnittger et al. 9 How ticks spread disease11 Ticks transmit pathogens that cause disease through the process of feeding. Depending on the tick species and its stage of life, preparing to feed can take from 10 minutes to 2 hours. When the tick finds a feeding spot, it grasps the skin and cuts into the surface. The tick then inserts its feeding tube. Many species also secrete a cement-like substance that keeps them firmly attached during the meal. The feeding tube can have barbs which help keep the tick in place. Ticks also can secrete small amounts of saliva with anesthetic properties so that the animal or person can’t feel that the tick has attached itself. If the tick is in a sheltered spot, it can go unnoticed. A tick will suck the blood slowly for several days. If the host animal has a bloodborne infection, the tick will ingest the pathogens with the blood. Small amounts of saliva from
Thyroid Function in Animals

Sushant Sadotra Introduction: The thyroid is one of the endocrine glands in vertebrates. The thyroid gland has a bilobed structure located below the larynx and overlays the trachea in animals. In different animals, Anatomical variations of the thyroid are primarily seen in the isthmus connecting the gland’s two lobes. The size of the gland approximates 0.20% of body weight. However, the size may increase due to iodine deficiency, ingestion of goitrogenic toxins, tumors, and hyperactivity of the gland, or maybe reduced to fibrotic due to hyperthyroidism. Thyroid follicles are the thyroid gland’s functional units with a spherical structure composed of an inner core of the thyroglobulin-hormone complex, colloid. The colloid is surrounded by an outer monolayer of follicular cells and acts as the storage tank of thyroid hormone. The overall size of the follicles and the shape of their follicular cells may differ due to the functional activity of the thyroid gland. The dormant follicular cells are squamous-shaped compared to the tall columnar, highly active follicular cells. Other than colloid, the thyroid C-cells are interspersed between the follicles. The thyroid C-cells are the source of the hypocalcemic hormone calcitonin that is associated with calcium metabolism. The third type of tissue embedded in the thyroid gland is the parathyroid. The parathyroid is the source of the hypercalcemic hormone parathormone. Functions of the thyroid gland: The thyroid gland functions the same in all animals. There are four primary functions of the thyroid gland in animals; trapping the iodide, synthesis of thyroid hormones, storage, and release of hormones. All these activities of the thyroid gland are usually regulated by thyroid-stimulating hormone (TSH), a pituitary hormone. Hormonogenesis and release of thyroid hormone mainly have four stages: Trapping of Iodide: The follicular cells trap the circulating I– from the blood against the concentration gradient mediated by a sodium iodide symporter (NIS) protein present on the thyroid follicular cell membrane. A trapping enzyme catalyzes the trapping process via a mode of active transport catalyzed by an ATP-dependent Na+K+-ATPase. This trapping system’s high efficiency can concentrate most blood iodine in the thyroid gland. This process is stimulated by TSH and blocked by large amounts of I– or goitrogenic agents. (Figure 1) Synthesis of Thyroid Hormones: The trapping of I- is followed by its oxidation, catalyzed by peroxidase to form a highly active free radical I*. This reaction is also stimulated by TSH and inhibited by thyrotoxic agents (thiouracils or thioureas). At the follicular cell membrane-colloid interface, highly active I* binds to thyroglobulin, a thyroidal glycoprotein of a high molecular weight of 660 kDa. I* binds to thyroglobulin at its tyrosine moieties to form monoiodotyrosine (MIT) or a diiodotyrosine (DIT). After that, the iodinated phenyl groups of the tyrosine undergo oxidative condensation resulting in the synthesis of thyroid hormones. The thyroid gland produces two active hormones: 3,5,3’-triiodothyronine (T3) and 3,5,3′,5,-tetraiodothyronine (T4). T3 is formed by the oxidative condensation of an iodinated phenyl group of one DIT to an MIT group or of one DIT to another DIT to create T4. The inner deiodination product of the T4 is the inactive hormone is 3,3′,5’-triiodothyronine (rT3). Storage of hormones: Thyroid follicular cells synthesized thyroglobulin and localized it to the cell membrane for the iodination process. Iodinated thyroglobulin, also known as a colloid, is released and stored in the lumen of the follicle. Release of hormone: TSH stimulates the release process of hormones. TSH acts at the follicular cell membrane, the second site of action for TSH. Colloid from the lumen is taken up to the follicular membrane, where they are taken in as vesicles into the follicular cells by the process of endocytosis. Lysosomes merge with these vesicles to release lysosomal proteases that hydrolyze the colloid. Hydrolyses of colloid release their T3, T4, DIT, and MIT. Microsomal tyrosine deiodinases enzymatically degrade the released DIT and MIT, and their iodine is recycled within the follicular cell. A simple diffusion process releases the T4 and T3 into circulation. Out of the total hormone released from the gland, 90% is T4, and only 10% is T3. The phenyl group of T4 may also undergo some deiodination within the gland or in the peripheral tissues to form rT3. rT3 is an inactive form of the T3 hormone. Therefore it undergoes a degradation pathway. (Figure 1) The regulation of T3 and T4 secretion starts in the hypothalamus. The thyrotropin-releasing hormone secreted from the hypothalamus acts on the pituitary gland. This stimulates TSH secretion, which ultimately acts on the thyroid gland, producing and releasing thyroid hormones. The action and disorder of Thyroid Hormones: In humans and animals, thyroid hormones play a vital role in regulating metabolic and cellular mechanisms. The mode of action can be quick in minutes or prolonged to hours or longer. Thyroid hormones in normal levels work together with other hormones like insulin (beta cells of the pancreatic islets) and growth hormone (pituitary gland) to work on protein synthesis in different cellular processes. However, thyroid hormones can be catabolic in excess (hyperthyroidism), with increased protein breakdown and gluconeogenesis. Hyperthyroidism is more common in cats middle-aged to old cats than in dogs. However, thyroid carcinoma could be a cause when it occurs in dogs. Decreased levels of thyroid hormones (hypothyroidism) cause a slower metabolic rate in animals. This disorder is most likely seen in middle-aged (4-10 Years) dogs and mid to large-size dog breeds (Doberman pinscher, Golden retriever, etc.). Also, spayed female dogs have a higher hypothyroidism risk than unspayed ones. On the contrary, naturally occurring hypothyroidism is rare in cats. Reference: Kaneko, J. J. (2008). Clinical biochemistry of domestic animals. San Diego: Academic Press. Thanas C, Ziros PG, Chartoumpekis DV, Renaud CO, Sykiotis GP. The Keap1/Nrf2 Signaling Pathway in the Thyroid—2020 Update. Antioxidants. 2020; 9(11):1082. https://doi.org/10.3390/antiox9111082 Mark E. Peterson. The Thyroid Gland in Animals. Last full review/revision Jul 2019 | Content last modified Oct 2020. MSD MANUAL Veterinary Manual, https://www.msdvetmanual.com/endocrine-system/the-thyroid-gland/the-thyroid-gland-in-animals
Boxer Dog Health Guide: Genetics, Disease Risks, and PCR Diagnostics

Table of Contents 1. Introduction Overview of the Boxer Breed The Boxer breed originated in late 19th-century Germany through the deliberate cross-breeding of the Bullenbeisser, a traditional large hunting breed, with the English Bulldog and other breeds. The goal was to produce an athletic, versatile working dog with a stable temperament. Historically used for hunting large game, Boxers were introduced to the United Kingdom in the 1930s and have since remained popular as both working and companion dogs. Physically, Boxers are medium-large, muscular dogs with a short coat and a moderately brachycephalic skull. Their distinctive features include a muzzle approximately one-third the length of the head, a broad upper jaw, an undershot lower jaw, and long, straight limbs. Common coat colors are fawn and brindle, with roughly 10% of Boxers being white. Modern Boxers are celebrated for their friendly, energetic, and curious temperament, although studies indicate that some may exhibit protective or guarding behaviors, and male Boxers may have a higher propensity for aggression, which should be monitored. Importance of Breed-Specific Disease Knowledge Awareness of breed-specific health risks is critical for owners, breeders, and veterinarians. Understanding the Boxer’s unique health profile enables the development of Breed Health and Conservation Plans, prioritizing conditions such as cancer, cardiac disorders, and Degenerative Myelopathy (DM). For breeders, this knowledge informs responsible breeding decisions that can reduce inherited disease prevalence. Owners and veterinarians benefit from early recognition of clinical signs, such as fainting or sudden death associated with Arrhythmogenic Right Ventricular Cardiomyopathy (ARVC), which can allow life-saving interventions before severe disease progression. Modern Diagnostic Tools Early detection and monitoring of Boxer-specific diseases have been greatly enhanced by modern diagnostic tools: DNA Testing: Enables identification of genetic risk factors. For Boxers, tests are available for ARVC1 and ARVC2 gene variants, as well as the SOD1A mutation linked to Degenerative Myelopathy, classifying dogs as clear, carriers, or at-risk. Holter Monitoring: A 24-hour Holter monitor is the gold standard for detecting ARVC, capturing intermittent ventricular premature complexes (VPCs) that may be missed in a standard ECG. Echocardiography with Doppler: Provides detailed visualization of heart structures and blood flow, essential for diagnosing Subaortic Stenosis (SAS) and other valvular conditions. 2. History and Origins The Boxer breed was developed in late 19th-century Germany through the deliberate cross-breeding of the traditional large hunting breed Bullenbeisser with English Bulldogs and other breeds. The goal was to produce an athletic working dog with a stable temperament. Originally bred for hunting and guarding, the Boxer was first officially registered in the United Kingdom in 1939 and has since evolved into a highly popular family companion, valued for both intelligence and character. Over time, specific physical and behavioral traits were emphasized to define the breed: Strength and Agility: Boxers are medium-large, muscular dogs with an athletic, square build, well-developed muscles, and high endurance. Playful Temperament: Known for being curious, lively, and energetic, Boxers retain their playful nature well into adulthood. They are also widely regarded as sweet-tempered and high-spirited, making them engaging companions for families. Loyalty and Trainability: Boxers are eager to please, respond well to consistent training, and thrive on positive reinforcement, often appearing proud and satisfied when praised. Courage and Protective Instincts: While naturally sociable, Boxers may demonstrate home-protective or guarding behaviors, such as alert barking toward strangers. These instincts are typically balanced by a fundamentally friendly and submissive nature with familiar people. While the breed retains its historical strength and agility, its modern role has shifted primarily to that of a beloved family dog, celebrated for its intelligence, energy, and affectionate personality. 3. Modern Diagnostic and Preventive Tools 1. Cardiomyopathy (Arrhythmogenic Right Ventricular Cardiomyopathy, ARVC) Also known as Boxer Cardiomyopathy, ARVC is an adult-onset, inherited disease affecting the heart’s electrical system. Pathophysiology: Normal myocardial tissue is gradually replaced by fatty or fibrous tissue, disrupting electrical conduction. Clinical Signs: May include syncope (fainting), exercise intolerance, lethargy, and sudden cardiac death. Less than 5% develop congestive heart failure. Genetics: Variants in ARVC1 (striatin gene) and ARVC2 are implicated, though the disease shows incomplete penetrance. Diagnosis: 24-hour Holter monitoring is the gold standard, as intermittent ventricular premature complexes (VPCs) may be missed on a standard ECG. Management: Anti-arrhythmic drugs (e.g., sotalol, mexiletine) are used alongside Omega-3 fatty acids and sometimes L-carnitine supplementation. 2. Aortic Stenosis (Subaortic Stenosis, SAS) SAS is a common congenital heart defect in Boxers. Description: Abnormal tissue below the aortic valve obstructs blood flow, causing the left ventricle to hypertrophy. Clinical Signs: Mild cases may be asymptomatic; moderate to severe cases can present with weakness, fainting, or sudden death. Breeding Considerations: Affected dogs should not be bred, as the condition is hereditary. Diagnosis: Confirmed via echocardiography with Doppler, allowing detailed visualization of obstruction and measurement of blood flow. 3. Bone and Joint Problems Boxers are susceptible to various musculoskeletal disorders. Hip Dysplasia: Screening via radiographs is recommended at ≥2 years of age for breeding candidates. Cranial Cruciate Ligament (CCL) Ruptures: Leads to instability and pain in the knee joint. Prevention and Management: Maintaining healthy weight, high-quality diet, and avoiding excessive strain is key. Supplements like glucosamine-chondroitin may aid joint health, particularly in older dogs. 4. Degenerative Myelopathy (DM) DM is a progressive, incurable neurological disorder affecting the spinal cord. Onset: Typically appears between 7–14 years of age. Signs: Early signs include knuckling, dragging rear paws, and hind limb weakness. Progression leads to paralysis of hind and eventually front limbs, urinary incontinence, and potentially compromised respiratory function. Genetics: Caused by a recessive SOD1 mutation; dogs can be clear, carriers, or at-risk. Management: No cure exists, but regular physical activity and assistive devices (e.g., wheelchairs, lifting harnesses) improve quality of life. 5. Cancer Neoplasia is a major health concern in Boxers. Predisposition: Boxers are particularly prone to Mast Cell Tumors (MCTs) and may also develop tumors of the brain, thyroid, mammary glands, testes, heart, spleen, and lymphoma. Prevalence: One study reported a 14.2% prevalence, higher than many other medium-large breeds. White Boxers: Dogs with white markings may be slightly more susceptible to
Hyperthyroidism is not a risk factor for subclinical bacteriuria in cats: A prospective cohort study
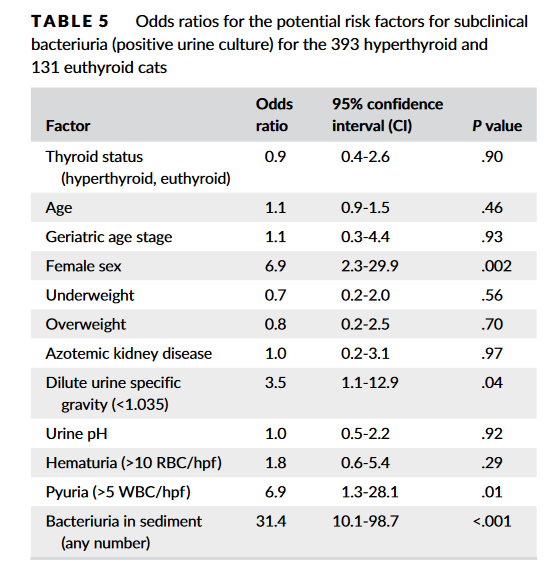
Source: https://doi.org/10.1111/jvim.15769 Subclinical bacteriuria is the presence of bacteria in urine with no clinical evidence of urinary tract infection (UTI). Some previous studies indicated that hyperthyroid cats are susceptible to UTIs (mostly subclinical) with prevalence rates of 12%-22%. As a result, many clinicians would recommend urine culture when evaluating hyperthyroid cats. To determine the true prevalence of subclinical bacteriuria in cats with hyperthyroidismIn, Eeterson M.E.’s group conducted a prospective cohort study. Both hyperthyroid and euthyroid cats had urine collected by cystocentesis for complete urinalysis and culture. Data pertaining to age, sex, body condition, and serum thyroxine and creatinine concentrations also were acquired. Hyperthyroid cats showed a low prevalence of subclinical bacteriuria (4.3%), which did not differ from that found in euthyroid cats (4.6%) in this study. In addition, only female sex was a significant risk factor. In conclusion, hyperthyroid cats are not at risk for subclinical bacteriuria.
Alaskan Malamute Guide: Health, Care, and Genetic Screening

Table of Contents 1. Introduction Overview of the Alaskan Malamute Breed The Alaskan Malamute is a powerful, heavy-duty spitz-type dog, historically bred as a freight-carrying sled dog in the Arctic. Its name derives from the Mahlemut Inuit tribe, who developed the breed to haul heavy loads across Alaska’s Norton Sound region. Known as the “Freight Train of the North,” Malamutes were indispensable partners for survival, assisting in hunting seals and polar bears, transporting goods, and providing warmth in tribal shelters. Frequently mistaken for Siberian Huskies, Malamutes are larger and more robust, designed for strength and endurance rather than speed. Their history as working dogs has shaped their physical abilities and behavioral traits, making them both highly capable and demanding companions. Breed-Specific Traits and Temperament Understanding the Malamute’s unique personality is essential for potential owners. They are highly intelligent but strong-willed and independent, combining loyalty, affection, and playfulness with a persistent “puppy-like charm” into adulthood. Their pack-oriented heritage means they require a confident, consistent leader to prevent dominance issues. Key behavioral traits: Vocalizations: Rather than typical barking, Malamutes are vocal and expressive, often howling, making “woo-woo” sounds, or “singing.” Exercise Needs: Extremely active, requiring a minimum of two hours of vigorous daily exercise; without adequate stimulation, they may become destructive or overly vocal. Prey Drive: High prey drive requires caution around smaller animals and can contribute to same-sex aggression. Grooming: Their thick double coat protects them in freezing climates but leads to heavy seasonal shedding, requiring daily brushing to maintain coat health. Health Needs and Veterinary Considerations Expert veterinary care is critical, as Malamutes are predisposed to several hereditary health conditions, many of which are autosomal recessive. Responsible breeding and early detection are key to maintaining a healthy population. Common health concerns include: Hip Dysplasia: Malformation of the hip joint that can lead to arthritis and reduced mobility; x-ray screening is essential for breeding dogs. Alaskan Malamute Polyneuropathy (AMPN): A neuromuscular genetic disorder causing weakness, muscle wasting, and a characteristic bunny-hopping gait; DNA testing identifies carriers and affected individuals. Day Blindness (Cone Degeneration): Sensitivity to bright light, causing difficulty seeing during the day while retaining night vision. Hypothyroidism: An underactive thyroid that can affect weight, skin, and energy levels. Gastric Dilation and Volvulus (GDV/Bloat): A life-threatening twisting of the stomach, most common in deep-chested breeds, requiring immediate veterinary attention. To reduce these risks, the Alaskan Malamute Club of America mandates testing for hip dysplasia, polyneuropathy, and ocular phenotypes in breeding dogs. Owners are advised to maintain a balanced, protein-rich diet while monitoring weight, as their thick coat can easily conceal obesity. Early socialization and consistent, reward-based training provide the foundation for a healthy, well-adjusted companion. 2. History and Origins The Alaskan Malamute is an ancient Arctic breed originating in Alaska’s Norton Sound region, where it was developed by the Mahlemut Inupiaq people. These dogs were indispensable partners for human survival, assisting in hunting polar bears and seals and providing warmth in tribal shelters during freezing nights. Bred to pull heavy sleds over vast, frozen landscapes, the Malamute earned the nickname “Freight Train of the North” for its exceptional strength and ability to haul freight over long distances. The breed’s reputation for resilience grew during the 1896 Gold Rush, and Malamutes later participated in notable historical missions, including Admiral Byrd’s Antarctic expeditions and World War II search and rescue operations. Their physical and behavioral traits, honed over centuries, allowed them to thrive in extreme Arctic conditions. The American Kennel Club (AKC) officially recognized the Alaskan Malamute in 1935, formalizing the preservation of the breed’s pure bloodlines and distinctive characteristics. While the sources do not explicitly mention the United Kennel Club (UKC), references frequently highlight the role of the Kennel Club (UK) and the importance of using Assured Breeders to maintain breed standards and promote health. To endure harsh Arctic climates, Malamutes evolved several adaptive traits: Insulating Double Coat: A dense, woolly undercoat paired with a coarse, water-repellent outer layer protects against freezing temperatures. Immense Strength: Unlike Siberian Huskies, which were bred for speed, Malamutes prioritize power and substantial bone, enabling them to pull loads multiple times their own body weight. Specialized Features: Erect ears enhance alertness, and the well-furred plumed tail, carried over the back, provides both a majestic appearance and protection from the elements. These adaptations, combined with their strong pack instincts and endurance, establish the Malamute as one of the most capable and iconic Arctic working breeds. 3. Physical Characteristics Size and Sexual Dimorphism Alaskan Malamutes are large, powerful dogs, with a standard height of 23 to 25 inches at the shoulder and a typical weight range of 75 to 85 pounds. The breed exhibits clear sexual dimorphism, with males generally larger and heavier than females. Adult males may reach 25 to 28 inches in height and weigh up to 85 pounds, while females typically measure 23 to 25 inches and weigh between 65 and 75 pounds. Some Malamutes from working lines can surpass these dimensions, reaching weights of up to 100 pounds, emphasizing the breed’s capacity for heavy-duty work. Coat Types and Seasonal Shedding The Alaskan Malamute is equipped with a specialized thick double coat adapted for survival in Arctic climates. This coat consists of: A soft, dense, woolly undercoat that provides insulation. A straight, harsh, water-repellent outer coat that stands off from the body and protects against moisture and wind. Due to this dense coat, Malamutes are heavy shedders year-round. They also experience pronounced seasonal shedding twice annually—spring and autumn—commonly referred to as “blowing” the undercoat. During these periods, daily or even twice-daily brushing is necessary to manage loose fur and prevent matting. Recognizable Markings and Differences from Huskies Distinctive physical features set Malamutes apart from other sled dogs: Tail: A well-furred, plumed tail carried over the back, both functional for warmth and aesthetically striking. Head and Ears: Erect, triangular ears, a broad head, and almond-shaped eyes contribute to an alert and expressive appearance. Coat Colors and Markings: Coat colors include gray, black, sable, and red, always
Predicting Outcomes in Hyperthyroid Cats Treated with Radioiodine
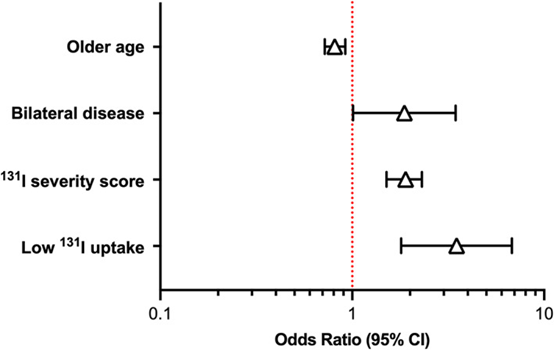
Source: J Vet Intern Med. 2022 Jan-Feb; 36(1): 49–58. doi: 10.1111/jvim.16319. Hyperthyroidism is a common disease in cats, and mostly afflicts cats middle-aged and older. It is caused by the overproduction of thyroid hormone by the thyroid glands. Radioiodine (131 I) is the treatment of choice to restore euthyroidism with a single dose of radiation without producing hypothyroidism for cats receiving the treatment. However, 30% to 50% of 131I‐treated cats develop iatrogenic hypothyroidism after treatment. Besides, 5% to 10% of hyperthyroid cats fail 131I treatment and remain persistently hyperthyroid. To identify pretreatment factors that may help predict persistent hyperthyroidism and iatrogenic hypothyroidism after treatment of cats using a novel 131I dosing algorithm. Peterson, ME and Rishniw, M conducted a study involving one thousand and four hundred hyperthyroid cats treated with 131I. Cats underwent an evaluation that included a complete physical examination, routine laboratory testing (CBC, serum biochemical profile, complete urinalysis), determination of serum thyroid hormone concentrations (total thyroxine [T4], triiodothyronine [T3], and thyroid‐stimulating hormone [TSH]), and qualitative and quantitative thyroid scintigraphy. Pretreatment predictors (clinical, laboratory, scintigraphic, 131I dose, 131I uptake measurements) of treatment failure or iatrogenic hypothyroidism were identified by multivariable logistic regression analysis. In conclusion, Age, sex, serum TSH concentration, bilateral and homogeneous 99mTc‐pertechnetate uptake on scintigraphy, severity score, and percent 131I uptake are all factors that might help predict outcome of 131I treatment in hyperthyroid cats. For detail information please check the original paper: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8783366/
Pug Health and Common Diseases: From Breed Traits to PCR Diagnostic

Table of Contents 1. Introduction The Pug is among the oldest recognized companion dog breeds, with origins tracing back more than two millennia to imperial China. Unlike many modern breeds shaped by functional roles such as hunting, herding, or guarding, the Pug was selectively developed almost exclusively for companionship and symbolic value within ruling households. This unique breeding trajectory has resulted in a dog whose physical form has long been governed by human aesthetic preference rather than biological performance. Historical records describe Pugs as highly prized animals within Chinese imperial courts, where they were bred for specific facial features associated with nobility and spiritual symbolism. As the breed spread to Europe through early global trade networks, these aesthetic traits were further refined and standardized. Over time, selection increasingly favored shortened muzzles, prominent eyes, and compact body conformation, characteristics now formally classified as brachycephalic. While these traits define the modern Pug’s appearance, they also underpin a distinct and well-documented disease profile. From a veterinary and molecular perspective, extreme craniofacial shortening alters airway anatomy, ocular protection, thermoregulation, and musculoskeletal biomechanics. As a result, Pugs exhibit a disproportionately high prevalence of conditions such as brachycephalic obstructive airway syndrome, corneal injury, dermatological disease associated with skin folds, and inherited neurological disorders. Understanding breed-related disease in the Pug therefore requires more than a catalog of clinical symptoms. It demands an appreciation of how historical selection pressures, genetic architecture, and modern breeding practices converge to influence health outcomes. This article examines the major inherited and conformation-associated diseases affecting Pugs, integrating veterinary medicine, genetics, and evidence-based clinical management to inform responsible ownership and breeding strategies. 2. Physical Characteristics and Breed Conformation Building on their history as imperial companions, the modern Pug is physically characterized by compact strength and a distinctive craniofacial structure. This combination is traditionally summarized by the Latin motto “multum in parvo”, meaning “a lot in a little,” reflecting a breed that is small in size yet substantial in build and presence. General Build and Proportions Despite their classification as a toy breed, Pugs are notable for their solid and muscular frame. Weight and Height: Adult Pugs typically weigh between 14 and 18 pounds and measure approximately 10 to 14 inches at the shoulder. Body Shape: The breed is described as square and cobby, indicating a short-backed, thickset body with good substance. Proportional Balance: Breed standards emphasize overall symmetry. Excessively long limbs, a narrow frame, or disproportionate body length are considered deviations from ideal conformation. Each anatomical component should contribute to a compact, well-integrated whole. Distinguishing Facial Features The Pug’s facial anatomy is its most recognizable feature and plays a central role in both breed identity and health considerations. Head: The skull is large, round, and massive, without doming or indentation. A flat skull profile is preferred over an apple-shaped contour. Eyes: Pugs possess very large, dark, and prominent globular eyes, producing an alert and expressive appearance. The orbits are relatively shallow, contributing to ocular prominence. Muzzle: The muzzle is short, blunt, and square, a conformation classified as brachycephalic, characterized by significant shortening of the facial bones. Dentition: A slightly undershot bite is typical, with the lower incisors extending marginally beyond the upper incisors. Facial Wrinkles: Deep and well-defined skin folds are a hallmark of the breed. Historically, forehead wrinkles resembling the Chinese character for “prince” (王) were selectively valued. Coat and Coloration The Pug’s coat reflects its development as a companion animal rather than a working breed. Texture: The coat is short, fine, smooth, and glossy, lying close to the body. Coarse or woolly textures are not characteristic. Accepted Colors: Recognized coat colors include fawn and black, with fawn ranging from pale silver to deep apricot tones. Markings: In fawn individuals, darker pigmentation forms a distinct black mask, black ears, and a dorsal trace, a narrow black line extending along the spine toward the tail. Conformation-Associated Health Considerations The defining physical traits of the Pug are closely associated with a breed-specific health profile. Brachycephalic Airway Changes: The shortened muzzle and compact nasal passages are associated with brachycephalic obstructive airway syndrome (BOAS), which can affect breathing efficiency and heat tolerance. Ocular Exposure: Prominent eyes and shallow orbits increase susceptibility to corneal injury, ulceration, and globe displacement following trauma. Skin Fold Conditions: Deep facial wrinkles may retain moisture and debris, creating a microenvironment conducive to intertrigo and secondary microbial overgrowth. Body Condition Sensitivity: The breed’s compact frame predisposes it to excess weight gain, which may exacerbate respiratory, orthopedic, and metabolic stress when not appropriately managed. 3. Temperament and Lifestyle Characteristics Pugs are defined by their even-tempered, affectionate, and sociable nature, often described as possessing a “multum in parvo” (a lot in a little) personality. They balance a playful, mischievous sense of humor with a dignified, stable temperament, making them one of the most cherished companion breeds. The “Shadow” Companion A hallmark of the Pug’s lifestyle is its absolute devotion to its human family: Constant Presence: Pugs are famously called “shadows” because they tend to follow their owners from room to room, craving physical closeness and attention. Emotional Intuition: They are highly sensitive to their owners’ moods and often respond with an eager-to-please attitude. Need for Interaction: Pugs thrive on human companionship and can become distressed if left alone for extended periods. Social Compatibility Pugs are known for their friendliness and ability to integrate seamlessly into diverse households: Children: They have a natural affinity for children. Unlike some delicate toy breeds, Pugs are sturdy enough to withstand supervised play and heavy-handed affection. Other Pets: Properly socialized Pugs generally get along well with other dogs and can coexist peacefully with cats and other small animals. Strangers: They are typically friendly toward newcomers and rarely show aggression, often greeting visitors with a wagging tail. The “Clown” Persona Pugs are often called the “clowns” of the canine world due to their expressive faces and entertaining antics: Mischief and Dignity: They combine playful antics with poise, amusing their owners while maintaining a sense of self. Willful Nature: Despite a strong
