Bird Sexing

Bioguard Corporation Many mammals are sexually dimorphic, which means that their sex can be identified based on their appearance. However, it is not the case for most birds. Many birds are sexually monomorphic although males and females may differ markedly in size, color, and appearance in some birds, such as peacocks (Fig. 1). Knowledge of a bird’s gender is important for veterinary practitioners, ornithologists and aviculturists. Gender identification in birds is beneficial for proper pairing of birds, and preparing owners for the eventual changes their bird will experience as it matures. Furthermore, knowing the gender of a bird allows veterinarians to diagnose gender-specific diseases. Traditional sexing The traditional methods used to identify a bird’s gender include behavioral observation, morphological differences, acoustic analysis, laparoscopic examination, laparotomy, vent sexing (cloacal examination), steroid sexing, and chromosome inspection (karyotyping). These methods can be inaccurate, invasive, or costly. As an alternative to these methods, molecular DNA-based sexing methods provide fast, accurate and non-harmful to the bird. DNA sexing The XY sex-determination system is used to classify many mammals, some insects (Drosophila), and some plants (Ginkgo tree). In this system, most females have two of the same kind of sex chromosome (XX), while most males have two distinct sex chromosomes (XY). The sex of birds is determined by the ZW sex determination system. Females have the heterogametic sex chromosomes ZW, whereas males have the homogametic ZZ. The standard method of molecular sexing in birds relies on PCR amplification of non-coding sequences (introns) located in the sex-chromosome-specific gene encoding the Chromo-Helicase-DNA binding protein (CHD1), and mapping on the sexual W (CHD-W) and Z chromosomes (CHD-Z). The length polymorphisms between CHD-W and CHD-Z alleles allows for sex discrimination. Amplification of the CHD gene leads to a single amplicon in males and two distinct amplicons in females. In addition to PCR, other PCR-based technologies have been applied to avian sexing, including Single Strand Conformation Polymorphism (SSCP), Restriction Fragment Length Polymorphism (RFLP), and Amplified Fragment Length Polymorphism (AFLP). These methods, however, are not viable for commercial use. Conclusion DNA sexing provides over 99.99% accuracy in determining a bird’s gender. This testing allows bird owners to quickly and accurately identify their birds’ gender, thereby enhancing the efficiency of pairing for breeding and conservation efforts.
Severe Fever with Thrombocytopenia Syndrome
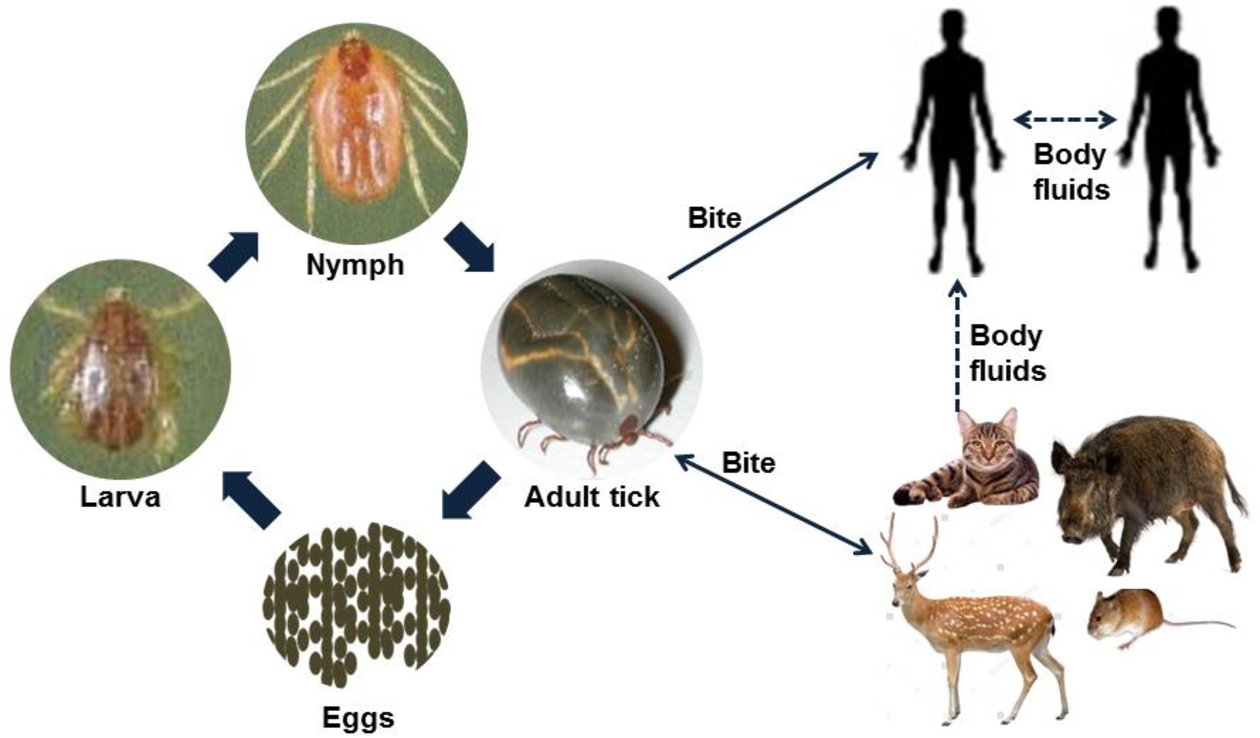
Bioguard Corporation Severe Fever with Thrombocytopenia Syndrome (SFTS) is an emerging infectious disease that was first reported in China in 2011. It is caused by the Severe Fever with Thrombocytopenia Syndrome virus (SFTSV) and is transmitted to humans and animals through the bite of an infected tick. The main clinical symptoms of SFTS include fever, vomiting, diarrhea, multiple organ failure, thrombocytopenia, leucopenia, and elevated liver enzyme levels. SFTSV has been detected in both domestic and wild animals, although most vertebrate animals were found to be sub-clinically infected with SFTSV. Pathogen SFTSV is a tick-borne virus belonging to the Genus Bandavirus (former Huaiyangshan Banyangvirus), Family Phenuiviridae. Vector and Disease Transmission The exact lifecycle and transmission mechanisms of SFTSV are not fully understood. However, ticks, particularly the Asian longhorned tick (Haemaphysalis longicornis), are believed to be the primary route of transmission. Other ticks, including Rhipicephalus sanguineus and Haemaphysalis concinna, have also been found to carry SFTSV. This suggests that ticks play a significant role in the transmission of SFTSV, similar to other members of the Phenuiviridae family, which are also known to be vector-borne. SFTSV Transmission Ticks are believed to be the primary vectors for SFTSV transmission, mainly through their bites. However, the virus can also be transmitted by infected animals or humans. Transmission of SFTSV from infected companion animals, such as cats and dogs, to humans has been reported through various routes. Pathogenesis Cytokine storm is thought to play a crucial role in the pathophysiology of SFTS. During the acute phase of the disease, the release of cytokines triggers a systemic inflammatory response. Histopathological examination of lymph nodes reveals necrosis and hemophagocytosis. The virus can be detected in the liver, spleen, adrenal glands, bone marrow, and lymph nodes. Thrombocytopenia may occur due to viral attachment to platelets, which are subsequently cleared by the spleen. SFTSV and Animal SFTSV has been detected in both domestic and wild animals; however, most vertebrate animals appear to be sub-clinically infected. Previous research indicates that antibodies (IgG/IgM) against SFTSV were found in goats and sheep (45.70%), cattle (36.70%), dogs (27.00%), chickens (9.60%), pigs (3.20%), and rodents (3.20%). A survey conducted in Taiwan identified SFTSV in Rhipicephalus microplus ticks collected from cows and goats, and antibodies against SFTSV were also detected in goats. In experimental studies, four out of six cats infected with SFTSV died, displaying symptoms as severe or more severe than those observed in human patients (Scientific Reports, 2019). Additionally, two captive cheetahs in a Japanese zoo died of SFTS in 2017, and there have been reports of fatal SFTSV cases in cats in Japan. References Casel MA, Park SJ, Choi YK. Severe fever with thrombocytopenia syndrome virus: emerging novel phlebovirus and their control strategy. Exp Mol Med. 2021 May;53(5):713-722. Seo JW, Kim D, Yun N, et al. Clinical Update of Severe Fever with Thrombocytopenia Syndrome. Viruses. 2021 Jun 23;13(7):1213. Sharma D, Kamthania M. A new emerging pandemic of severe fever with thrombocytopenia syndrome (SFTS). Virusdisease. 2021 Jun;32(2):220-227.
Cryptosporidiosis

Bioguard Corporation Cryptosporidiosis is an illness you get from the parasite Cryptosporidium. It causes watery diarrhea and other gastrointestinal (gut) symptoms. In addition to stomach infection, this parasite can infect the respiratory system causing a cough and/or problems breathing. The family Cryptosporididae belongs to the phylum Apicomplexa characterized by an anterior (or apical) polar complex (with apical rings, micronemes, and subpellicular microtubules), which allows penetration into host cells. Cryptosporidium species are able to infect a broad range of hosts including humans, domestic and wild animals (mammals, birds, fish, marsupials, reptiles, and amphibians) worldwide. Transmission and Life Cycle Humans and animals become infected with Cryptosporidium by touching anything that has come in contact with contaminated feces, although the most common mode of transmission is represented by ingestion of oocysts in contaminated food and water or air. Cryptosporidium has three developmental stages: meronts, gamonts, and oocysts. They reproduce within the intestinal epithelial cells. Two types of oocysts, thick-walled and thin-walled, are produced during sexual reproduction. Thick-walled oocysts are excreted from the host into the environment, whereas thin-walled oocysts are involved in the internal autoinfective cycle and are not recovered from stools. Oocysts are infectious upon excretion, thus enabling direct and immediate fecal-oral transmission. Clinical Symptoms The most common symptoms of cryptosporidiosis are watery diarrhea and stomach cramps. Other symptoms may include fever, nausea, vomiting, and loss of appetite. Symptoms and severity of infection vary with the age and immune status of the host. Cryptosporidium infections are uncommonly detected in cats and dogs. Cryptosporidiosis can sometimes make dogs and cats sick, but animals with signs are atypical. In most cases, epithelial damage is minimal, but in severe cases, infection is associated with losing the ability to maintain water balance. Clinical signs are usually restricted to mild diarrhea unless the host is immunosuppressed or has another underlying condition such as viral infection or malignancy. Diagnosis Cryptosporidiosis is a diarrheal disease that is spread through contact with the stool of an infected person or animal. The disease is diagnosed by examining stool samples. Oocyst excretion is intermittent, and multiple stool samples may be needed. Diagnostic methods include: Microscopic examination: Typically, stool samples are analyzed microscopically using various techniques, including acid-fast staining and Ziehl-Nielsen staining. Real-time PCR: The most accurate method for detecting Cryptosporidium spp. is through a fecal PCR assay. Immunologic tests: These include direct fluorescent antibody tests and enzyme immunoassays to detect Cryptosporidium sp. antigens. Treatment and Prevention Most patients with healthy immune systems will recover from cryptosporidiosis without treatment. Supportive measures, oral or parenteral rehydration, and hyperalimentation may be needed for immunocompromised patients with severe disease. The best way to prevent the spread of Cryptosporidium at home is by practicing good hygiene. References Sardinha-Silva A, Alves-Ferreira EVC, Grigg ME. Intestinal immune responses to commensal and pathogenic protozoa. Front Immunol. 2022 Sep 16;13:963723. Sponseller JK, Griffiths JK, Tzipori S. The evolution of respiratory Cryptosporidiosis: evidence for transmission by inhalation. Clin Microbiol Rev. 2014 Jul;27(3):575-86. Watier-Grillot S, Costa D, Petit C, et al. Cryptosporidiosis outbreaks linked to the public water supply in a military camp, France. PLoS Negl Trop Dis. 2022 Sep 12;16(9):e0010776.
Ferret Coronavirus

Bioguard Corporation The coronavirus of ferrets was first described in 1993. This ferret enteric coronavirus (FRECV) caused an enteric disease called epizootic catarrhal enteritis (ECE) or green slime disease. Later, a ferret systemic coronavirus (FRSCV)-associated disease, resembling the dry form of feline infectious peritonitis (FIP), was identified. ECE is characterized by high morbidity (nearly 100%) but low mortality (< 5%). FRSCV is associated with a high mortality rate and short duration of illness. Pathogen Coronaviruses are large, enveloped, positive-stranded RNA viruses classified under the genus Coronavirus within the family Coronaviridae, order Nidovirales. Virions are roughly spherical and are notable for the large spike (S) glycoprotein that extends from the virus envelope, which resembles a crown or solar corona when imaged using an electron microscope. The diameter of ferret coronavirus is about 120 nanometers, and its genome is about 28 kilobases. Transmission Both the enteric (FRECV) and systemic (FRSCV) ferret coronaviruses are classified as group 1 coronaviruses (that is, alphacoronaviruses), related to feline coronavirus and canine enteric coronavirus. The routes for transmission of FRECV and the FRSCV are suggested to be fecal–oral. Many facts of the pathogenesis of the virulent systemic ferret coronavirus remain unknown, but similar to FIPV, macrophages appear to play an important role in the inflammatory response. Clinical signs After infection of FRECV, the virus causes blunting of the intestinal villi and consequent maldigestion and malabsorption. Clinical signs include anorexia, vomiting, diarrhea, melena, dehydration, lethargy, and weight loss. The microscopic lesions include diffuse lymphocytic enteritis, with villus atrophy, fusion, and blunting; vacuolar degeneration and necrosis of the apical epithelium; or a combination of all these lesions. FRSCV-associated disease causing FIP-like lesions has been reported in mostly young (<18 months) ferrets. Clinical signs are nonspecific, including anorexia, weight loss, diarrhea, and enlarged intra-abdominal and, less commonly, peripheral lymph nodes. FRSCV is grossly associated with pale to white nodules (granulomatous inflammation) in multiple organs, including the spleen, kidneys, mesenteric lymph nodes, intestines, liver, lungs, and brains. Granulomas have a heterogenous cellular composition including macrophages, T and B lymphocytes, and plasma cells. Diagnosis Definitive diagnosis requires intestinal biopsy and histopathological examination, with confirmation of viral antigen or nucleic acid by immunohistochemistry or in situ hybridization. RT-PCR and electron microscopy can be used to examine feces from ferrets. Systemic coronavirus disease in ferrets can be confirmed by histological evaluation of biopsies and intralesional coronavirus nucleic acid detection. RT-PCR has been used to identify viruses in tissues and can differentiate ferret enteric coronaviruses from ferret systemic coronaviruses. References 1. Haake C, Cook S, Pusterla N, et al. Coronavirus Infections in Companion Animals: Virology, Epidemiology, Clinical and Pathologic Features. Viruses 2020, 12(9), 1023. 2. Osborne AJ, Hussain SS, Helman EE, et al. Ferret Systemic Coronavirus in Alpha-1 Antitrypsin Knockout Ferrets. Comp Med. 2022 Dec 1;72(6):410-415. 3. Gnirs K, Quinton JF, Dally C, et al. Cerebral pyogranuloma associated with systemic coronavirus infection in a ferret. J Small Anim Pract. 2016 Jan;57(1):36-9.
Infectious Myxomatosis of Rabbits
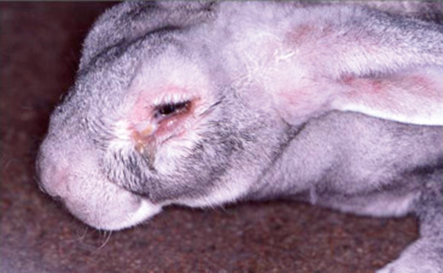
Bioguard Corporation Myxomatosis is primarily a disease of rabbits caused by infection with the myxoma virus. It mainly occurs in domestic and wild rabbits. The virus is harmless to humans. Myxomatosis can result in lumps developing around the ears and face. These lumps are named myxomas and the disease virus was named after this lesion. It was first discovered in South America, California and Mexico in 1896. Sick animals will die within a few days to two weeks after infection, and the fatality rate is close to 100%. Currently, there is no effective treatment. Pathogen Myxoma virus is the type species of the Leporipoxviruses, a genus of Chordopoxvirinae, double stranded DNA viruses, whose members infect leporids and squirrels, inducing cutaneous fibromas from which virus is mechanically transmitted by biting arthropods. Pathogenesis studies confirm that the virus initially replicates in dermal cells at the inoculation site, likely dendritic cells. From there, the virus spreads to local macrophages and epidermal cells, and to the draining lymph node. Virus replication in the latter results in lymphoid depletion, with extensive loss of cortical and paracortical lymphocytes. From the lymph node the virus spreads via blood leukocytes to distal tissues including the spleen and other lymphoid tissues, testis, lungs, and skin. Transmission and Clinical Signs Currently, myxoma virus is enzootic to the Americas, Europe, Australia and other regions. The principal mode of transmission of the virus is mechanical transport of virus on mouth parts by arthropod vectors such as ticks, mosquitoes, and fleas through bites. It can also transmit the virus to other rabbits via direct contact. The general incubation period is 3-7 days, up to 14 days. The first sign of disease is conjunctivitis that rapidly becomes more severe and is accompanied by a milky discharge from the eye. The rabbit has no energy and no appetite, with a fever that may reach 42°C. In severe outbreaks, some rabbits die within 48 hours after signs appear. Those that survive become progressively weaker and develop a rough coat. The eyelids, nose, lips, and ears become puffy, which gives a swollen appearance to the head. The ears may droop. In females, the vulva becomes inflamed and swollen with fluid; in males, the scrotum swells. Other signs include discharge of pus from the nose, difficulty breathing, and coma. Death usually occurs within 1 to 2 weeks after signs appear, and the fatality rate is close to 100%. Diagnosis Gross lesions: The most prominent gross lesions in in rabbits with myxomatosis are the skin tumors and the pronounced cutaneous and subcutaneous edema, particularly in the area of the face and around body orifices. Hemorrhages of the skin, heart, and subserosa of the gastrointestinal tract may be observed. Microscopic lesions: Lesions in the skin involve epithelial cells, fibroblasts, and endothelial cells and range from proliferative to degenerative, depending on the strain of virus. The skin tumors result from proliferation of undifferentiated mesenchymal cells, which become large stellate (myxoma) cells surrounded by a homogeneous matrix of mucinous material interspersed with capillaries and inflammatory cells Lab tests include serology and molecular diagnosis. Serology: immunofluorescent assay (IFA), ELISA, complement fixation test (CFT) Molecular diagnosis: PCR Treatment and Vaccination Unfortunately, there is no specific treatment for myxomatosis. Vet can only offer supportive care, including fluids, antibiotics to prevent secondary infections, and pain medication. The myxomatosis vaccine is available in some countries. Reference Bertagnoli S, Marchandeau S. Myxomatosis. Rev Sci Tech. 2015 Aug;34(2):549-56. Espinosa J, Ferreras MC, Benavides J, et al. Causes of Mortality and Disease in Rabbits and Hares: A Retrospective Study. Animals (Basel). 2020 Jan 17;10(1):158.
Diagnosis of Feline Respiratory Mycoplasma Infection

Bioguard Corporation In cats, ’mucosal’ mycoplasma infections typically cause ocular and respiratory disease, and less frequently neurological or joint disease. These Mycoplasma species are distinct to the haemotropic mycoplasmas that target red blood cells, causing hemolytic anemia in cats. Mycoplasma felis is typically associated with Upper Respiratory Tract Disease (URTD) in cats. Transmission M. felis is mainly transmitted from an infected cat to an in-contact one by aerosol, but also by grooming. Stresses, including overcrowding environments, concurrent respiratory viral infections, and poor hygienic situations, may promote transmission of the infection between cats. Clinical symptoms Mycoplasma felis is typically associated with URTD but sometimes it may be associated with lower respiratory tract infections. Common clinical signs include clear or colored discharge from the eyes or nose, coughing, sneezing, conjunctivitis, chemosis, lethargy, and anorexia. Lower respiratory tract infections can result in pneumonia with fever, cough, tachypnoea, and lethargy. Diagnosis Culture of mycoplasmas can be used to demonstrate infection, but it takes time for culture and rapid transport of samples to the laboratory is required. Demonstration of organisms via real-time PCR is increasingly being used to circumvent the difficulties with culture, Treatment Antimicrobial therapy is commonly used to treat mycoplasma respiratory infections. Doxycycline is a good first line agent because it is well tolerated by cats and relatively narrow in spectrum. The recommended dose is 5 mg/kg, PO, q12h or 10 mg/kg, PO, q24 (Lappin et al., 2017). Oxytetracycline or chlortetracycline ophthalmic ointment can be used q6h in addition as topical treatment. References: Vekšins A. Feline upper respiratory tract disease – Computed tomography and laboratory diagnostic. Vet World. 2022 Jul;15(7):1880-1886. Framst I, Ramesh P, Cai HY, Maboni G. Complete genome sequences of Mycoplasma cynos and Mycoplasma felis isolated from dogs and cats with infectious respiratory disease. Microbiol Resour Announc. 2024 Apr 11;13(4):e0124323.
Tritrichomonas Infection in Cat
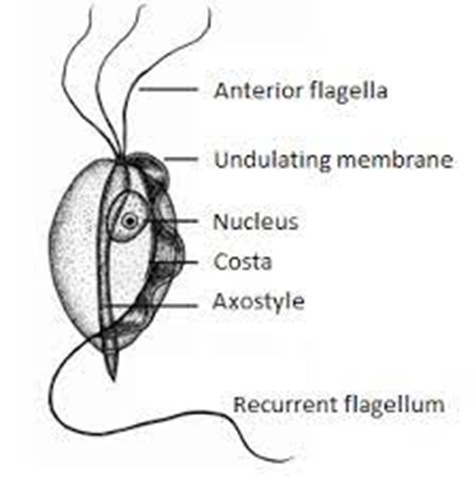
Bioguard Corporation Tritrichomonas foetus is a significant cause of large bowel diarrhea and persistent colitis in cats. These pear-shaped organisms have three anterior flagella and one posterior flagellum. They have a distinctive undulating membrane, which gives them a similar appearance to Giardia. However, they do not form cysts and are transmitted directly from one host to another as trophozoites. The infection is most prevalent among young cats living in close quarters, such as in densely populated catteries and shelters. A notable investigation into purebred show cats discovered a 31% infection rate among 117 cats spanning 89 catteries, as detailed in Gookin’s 2004 study Clinical Signs Some cats infected with T. foetus may not exhibit any symptoms, particularly older cats that are in good health. However, most cats with T. foetus infection suffer from mild to severe lymphoplasmacytic and neutrophilic colitis, which causes recurrent episodes of large bowel diarrhea that may vary in consistency from semiformed to “cow pie” and emit a foul odor. Diarrhea may contain fresh blood or mucus. Insevere cases, kittens may experience painful anal irritation, fecalincontinence, or even rectal prolapse. Affected cats usually maintain a healthy appearance and good body condition overall. The presence of diarrhea can be exacerbated by other intestinal infections or parasites, particularly Giardia and ryptosporidium. Diagnosis To confirm the infection of T. foetus, there are three methods available – direct fecal microscopy, fecal culture, and fecal polymerase chain reaction (PCR) assay. Direct fecal microscopy involves identifying the motile trophozoites T. foetus in fresh wet smears of diarrheic feces taken directly from the rectum. This method identifies the organisms in about 14% of cases and is less effective with formed or dried feces. In cats who have been treated with antibiotics recently, the detection rate decreases. Trichomonads, which resemble Giardia in size and shape, can be distinguished by their unique undulating membrane and rapid, jerky motility, contrasting with Giardia’s “falling leaf” movement. Fecal culture can be done in-house or at a specialized lab. It helps increase the chances of identifying the organisms. In specific cases, a saline flush might be performed by inserting a catheter through the cat’s anus to wash the colon with saline, followed by aspiration of fecal material. Fecal PCR is the most accurate method for identifying T. foetus. To perform this test, the fecal sample should not contain any litter. This technique detects the organism’s DNA traces in the cat’s stool. It’s best to conduct testing on cats that have not received antibiotics for at least two weeks for the most accurate test results. Antibiotics can temporarily reduce the number of T. foetus, leading to false negatives. Treatment Often, many approaches for treating chronic diarrhea have been tried unsuccessfully before a true diagnosis of T. foetus is confirmed. Tritrichomonas foetus is resistant to most antibiotics and is extremely difficult to eradicate (Gookin 2001). Numerous antibiotics have been evaluated. Some antibiotics reduce the number of organisms and improve the diarrhea without eliminating the infection, so diarrhea relapses whenever antibiotics are stopped. Diarrhea is typically refractory to corticosteroids. The most successful treatment for eliminating T. foetus is ronidazole (30 mg/kg PO, once or twice daily for 14 days). The side effects in some cats include lethargy, decreased appetite, and neurotoxicity. Cats with neurotoxic signs usually improve when the drug is stopped, but recovery can take 1 to 4 weeks. Ronidazole should not be used in pregnant and nursing queens or in very young kittens. Ronidazole is not approved for veterinary or human use, but some pharmacies compound chemical grade ronidazole for veterinary use. Because of its bitter taste, ronidazole compounded in gel caps is better tolerated than flavored suspension. When prescribing ronidazole obtain informed consent and instruct owners to wear protective gloves when handling it. Management of Tritrichomonas foetus Infection In cases where cats show mild or sporadic symptoms of diarrhea caused by T. foetus, and treatment is not possible due to potential side effects, costs, or the owner’s preferences, it is important to know that diarrhea may naturally go away with time, which can take up to two years. However, such cats are likely to remain lifelong carriers of the parasite. The outlook for cats receiving treatment is generally positive. A majority of treated cats exhibit better stool consistency in just a few days, though diarrhea might linger briefly as related secondary inflammation subsides. Nonetheless, around 25% of cases might experience a continuous infection despite initial treatment. Fortunately, T. foetus has a short lifespan outside its host and is easily neutralized by common disinfectants. To mitigate infection risks, it’s advised to uphold strict litter box cleanliness through daily cleansing, isolate cats under treatment, minimize stress factors, prevent overcrowding, and implement regular screenings in breeding and shelter settings whenever feasible.
Respiratory Tract Disease Complex in Cats

Sushant Sadotra, PhD/Diagnostic specialist Feline respiratory disease (FRD) syndrome or feline upper respiratory tract disease complex is a common infection in cats caused mainly by Feline Herpesvirus (FHV-1), Feline Calicivirus (FCV), Chlamydophila felis, Mycoplasma spp., and Bordetella bronchiseptica. About 90% of all upper respiratory infections are caused by FHV-1 and FCV. Common Symptoms: · Sneezing · Nasal congestion · Conjunctivitis (inflammation of the membranes lining the eyelids) · Discharge from the nose or eyes (clear, purulent, or cloudy containing pus). · Difficulty breathing · Ulcers in the mouth Less specific symptom · Less appetite · Lethargy · Fever · Enlarged lymph nodes · Blepharospasm (squinting) Sources of infection: · Susceptible cats can get an infection by contagious particles in saliva or secretions from the nose or eyes shredded by an infected cat. · Most cases are associated with direct contact · Natural transmissions can also occur via aerosol droplets. Stress may also cause a secondary course of illness. Real-Time PCR for Diagnosis: A definitive diagnosis is based on clinical signs and laboratory testing for the isolation and identification of the infection. The Polymerase chain reaction (PCR) test is one of the sensitive tests and most reliable for detecting the presence of infectious agents. PCR detects the genomic material of the pathogen and determines its presence in the host. It is often more sensitive and specific than other available tests. However, false negative results are still expected. In the case of patients with latent herpes infections where the FHV-1 is found in the trigeminal ganglion can give negative PCR results. In the case of Chlamydophila, 2-3 days of antibiotic treatment for patients can also have negative PCR results. Samples of ocular, nasal, or caudal pharyngeal secretions for PCR assay are best for the diagnosis and identification of causative agents. Sample collection tips: · Ocular: Moisten with tears/exudate well or firmly swab both of the conjunctival sacs with a sterile swab. · Clinical lesions: Prefer to swab from the nasal and pharyngeal areas. After sample collection, place the swab into the preservation buffer and mix thoroughly. If not for immediately use, please keep the mixture at 4°C (no more than 3 days) until nucleic acid extraction.
Feline Herpesvirus Infection- Diagnosis

Trinh Mai Nguyen Tang Feline herpesvirus-1 (FHV-1) is a feline respiratory infection virus also known as feline viral rhinotracheitis (FVR) [1]. The Herpes virus was first isolated by scientists Crandell and Maurer in 1958 in cats with respiratory infections [2]. This virus has a prominent genome with large double stranded DNA, belonging to the family Herpesviridae [3]. This virus is characterized by cat-to-cat transmission with an exposure rate of up to 97% [4]. Herpes virus can be inactivated at 37oC around 3 hours or at 56oC in 5 mins. Meanwhile, the virus can remains infective in the enviroment approximately 5 months and a month at 25oC [5]. Once a cat is infected with the herpes virus, it is incredibly difficult to completely treat it since the virus can enter a dormant state and continue to survive in the cat for the remainder of its life [6]. Cats are not infectious during this latent period, but if they are sick or going through a stressful period, the virus may be reactivate. If this occurs, the cat will once more get the infection and may represent symptoms [7]. Herpes viruses can be latent in the ganglion, attach to sensory nerves and reach nerve cells, persist in the nucleus of infected nerve cells and do not replicate, leading to the process of detecting this virus becomes difficult [7-8]. As reported by Ngoc.N.T and her colleagues, herpes virus can infect cats of any age, however, kittens are more susceptible [9]. Specifically, the prevalence of virus infection in cats younger than 6 months old, 6-12 months old and over 12 months old were 52.17%, 33.33% and 19.05%, respectively [9]. Although previous reports have demonstrate that gender has no effect on the incidence of herpes virus infection [7-8], but Henzel et al. (2002) found that isolates from female cats are substantially taller than isolates from male cats [10]. Clinical Symptoms Herpes virus enters the cat’s body by contact with infected tears, nose, saliva, or items, and then multiplies rapidly in the epithelium of the nose, nasopharynx, and conjunctival mucosa leading to primary infection [11]. In cats infected with FHV-1, signs of sadness, moodiness, lethargy, sneezing, fever, and discharge from the eyes and nose have been noted (figure 1-A), a process that is frequently extended 2-4 days or longer, depending on the immunological system of the cat [11]. Secondary infection occurred after the fourth day of incubation, with symptoms of infection in the throat, bronchi, and bronchioles, and the nasal and conjunctival epithelium necrosing [12]. Conjunctivitis is a common herpes virus symptom, indicated by congestive and exudative symptoms that develop over many days to purulent discharge (figure 1-B) [13]. Gaskell and Dawson (1988) found lung infection or bronchitis in cats, with kittens dying from pneumonia at a greater incidence than adult cats [11,14]. Some other atypical symptoms such as mouth and skin ulcers, dermatitis or neurological signs are rarely seen [7]. Furthermore, the mean white blood cell (WBC) count of cats infected with FHV-1 (17.77 ± 0.70 x103/μl) was slightly increased compared with that of normal cats (4.6-12.8 x 103/μl) [9], in which neutrophils, eosinophils and monocytes all showed signs of slight increase compared. Secondary infections of the eyes, upper respiratory tract, and necrotic ulcers of the mouth can all cause high white blood cell counts [11]. Table 1 displays the white blood cell count. Table 1. Hematological results of cats was infected with FHV-1 [9]. Targets Unit Reference ± SE Red blood cells ´ 106/μl 7-10,7 10,20 ± 0,64 Hemoglobin content g/dl 11,3-15,5 13,47 ± 0,54 RBC mass % 33-45 38,78 ± 1,41 Average volume of red blood cells fl 41-49 45,16 ± 0,75 Average amount of hemoglobin in red blood cells pg 14-17 15,57 ± 0,24 Platelet count ´ 103/μl 180-680 362,50 ± 30,82 WBC count ´ 103/μl 4,6-12,8 17,77 ± 0,70 Lymphocytes ´ 103/μl 1,05-6,00 4,50 ± 0,36 Mono leukocytes ´ 103/μl 0,05-0,68 0,96 ± 0,13 Neutrophils polymorphonuclear leukocytes ´ 103/μl 2,32-10,01 11,47 ± 0,45 Eosinophils ´ 103/μl 0,1-0,6 0,78 ± 0,08 Basophils ´ 103/μl 0-0,14 0,07 ± 0,01 Laboratory Diagnosis In the laboratory, there are many different methods used to determine FHV. Common approaches include PCR, virus isolation in cell culture, and indirect fluorescent antibody staining of tissue samples for viral antibody detection [8,16]. PCR FHV is one of the most common causes of upper respiratory tract illness in cats. Infected cats would show upper respiratory signs. Co-infection of FHV with other pathogens makes the clinical signs more severe, particularly feline calicivirus, Chlamydophila felis, Bordetella pneumoniaseptica, Mycoplasma species, Staphylococcus spp., or Escherichia coli [6]. When a cat is suspected of having a viral infection, a swab can be used to collect nasal, ocular, oropharyngeal secretions, corneal debris, aqueous humor, corneal samples, blood, or biopsies. By amplifying viral DNA, tPCR can detect genetic material of FHV in specimen. However, if the cat is not in the infectious phase, no virus particles will be shed, rendering the PCR test ineffective [19-20]. ELISA method – detecting IgG antibodies The enzyme-linked immunosorbent test (ELISA) is used to determine IgG antibodies against FHV or FHV-1 utilizing serum, aqueous humor, and cerebrospinal fluid samples [15]. This approach, however, cannot discriminate between diseased and vaccinated cats. Due to the extended latent period of FHV, the cat’s body will generate antibodies to combat it [8]. These neutralizing antibodies manifest 20-30 days after the first infection. As a result, the presence of antibodies in the serum signals a prior infection but does not always correspond with clinical signs [8]. Immunofluorescent antibody assay Another approach for detecting FHV is immunofluorescence antibody (IFA) testing on corneal or conjunctival smears or biopsiengs. Through an antigen-antibody response, this assay may identify viral proteins produced in cells. However, this approach is thought to be less sensitive than viral isolation or PCR [16]. Virus isolation This is a traditional method that can detect viruses through isolation of conjunctival debris, nose, oropharynx, or postmortem lung samples from infected cats [8]. This traditional method can detect
Canine Lyme Disease
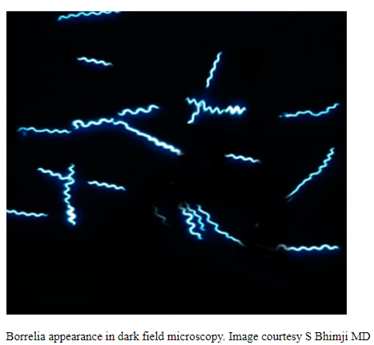
Oliver Organista, LA Lyme disease is a disease caused by the bacterium Borellia burdorgferi; a worm like, spiral-shape bacterium of spirochete class in the genus Borellia. The bacterium B. burgdorferi is transmitted through a bite of infected blacklegged tick or deer tick (Ixodes scapularis) to dogs and humans[1]. Different life-stage of I. scapularis ticks emerge at different times of the year (varies according to geographic location), giving a seasonality to Lyme disease transmission dynamics. It appears primarily in specific areas including the southern New England states; eastern Mid-Atlantic states; the upper Midwest, particularly Wisconsin and Minnesota; and on the West Coast, particularly northern California in the United States. It is also present in Europe and Asia[7]. Most of the areas where to find them are in forest or grassy, wooded, marshy areas near rivers, lakes or ocean, and are common in homes and buildings in secluded or rural areas. In Canada, there 2 types of blacklegged or deer tick that can spread Lyme disease. The blacklegged tick (Ixodes scapularis) and the blacklegged (Ixodes pacificus) [3] . Dogs tend to be bitten by infected I. scapularis adults, which are most active in the cooler early spring and late fall months [2]. An adult female tick is rarely (if ever) transmitted the B. burgdorferi to her offspring. Ticks most commonly become infected as juveniles after a bloodmeal on an infected wildlife host (most commonly rodents). Because ticks typically feed only one time per life stage, the next opportunity for B. burgdorferi transmission is during the next bloodmeal in the tick’s next life stage[2]. Typically, Lyme disease symptoms will take a couple of months or more to appear (2-5 months) after getting infected [8]. Symptomatically, Lyme disease can be difficult to distinguish from anaplasmosis because the signs of the diseases are very similar, and they occur in essentially the same areas of the country. Lyme disease is diagnosed through a blood test that shows whether an animal has been exposed to the bacterium[11]. Common symptoms that will appear are: Lameness: An inability to use one or more limbs is one of the most common symptoms of Lyme disease in dogs. Swollen lymph nodes: found in the neck, chest, armpits, groin, and behind the knees, are typically the first to show swelling. Lymph node swelling indicates an immune response triggered to fight the disease. Joint swelling: Swollen joints, stiff walking, or avoidance to touch may be other signs of the disease. Fatigue: Dogs with Lyme disease may also exhibit flu-like symptoms of low energy and lethargy. Loss of appetite: Losing interest in eating, especially if it leads to weight loss, is another sign that a dog may have Lyme. Fever: In addition to the above symptoms, a dog may have a fever caused by the Lyme disease infection. In rare cases, if Lyme disease is left untreated it can lead to damage in the kidneys, nervous system, and heart. Lyme disease affecting the kidneys is the second most common syndrome in dogs and is generally fatal. Facial paralysis and seizure disorders have been reported in the disease form affecting the nervous system. The form of the disease that affects the heart is rare. [10]. The most commonly used to diagnose Lyme disease in dogs are the serologic assays. Although some laboratories still use traditional serologic methods (e.g., whole-cell enzyme-linked immunosorbent assay and immunofluorescence assay), these assays have largely been replaced by serologic assays that detect host antibodies to specific B. burgdorferi proteins. These assays are qualitative, providing a yes/no answer regarding B. burgdorferi serostatus[3]. Treatment is generally recommended for seropositive dogs that display clinical signs of Lyme disease or are asymptomatic but have evidence of protein-losing nephropathy[4]. Most frequently antibiotics used to treat Lyme disease in dogs are doxyclycline and monicycline, at a dosage of 10mg/kg PO q12h to q24h for 30 days [2][5]. Amoxicillin and erythromycin are other antibiotics that can be used for treating the disease. A non-steroidal anti-inflammatory (carprofen or deracoxib) may also be given to the patient [6]. A possible complications may occur when treating Lyme disease. Some dogs who take antibiotics can develop loss of appetite, vomiting and diarrhea. Once infected, a dog will always have the bacteria that cause Lyme disease in his or her body. Therefore, relapses are possible; lookout for unexplained fever, swollen lymph nodes, and/or lameness. A small percentage of dogs develop kidney failure as a result of Lyme disease. Clinical signs include vomiting, weight loss, poor appetite, lethargy, increased thirst and urination, and abnormal accumulations of fluid within the body. [6] The best way to protect from Lyme disease is to use tick-preventive products year-round. Several safe and effective commercial parasiticides are available for tick control on dogs and cats, including systemics (isoxazolines), topicals (permethrin, fipronil), and collars. Another effective strategy is vaccination. Other prevention strategies include reducing exposure to ticks and avoiding areas with ticks [2]. References [1] Lyme Disease Diagnostic Market – Growth, Trends, COVID-19 Impact, and Forecasts (2022 – 2027), MOdor Intelligence, January 202 [2] Lyme Disease in Dogs: Signs and Prevention, Kathryn E. Reif, MSPH, PhD.,April 2020, https://todaysveterinarypractice.com/parasitology/lyme-disease/ [3] Lyme Disease, IPAC (https://ipac-canada.org/lyme-disease.php) [4] / Littman MP, Gerber B, Goldstein RE, et al. ACVIM consensus update on Lyme borreliosis in dogs and cats. J Vet Intern Med 2018;32(3):887-903. [5] Mullegger RR. Dermatological manifestations of Lyme borreliosis. Eur J Dermatol. 2004 Sep-Oct;14(5):296-309. PMID: 15358567 [6] How to Treat Lyme Disease in Dogs, Jennifer Coates, DVM, December 2014, PETMD (https://www.petmd.com) [7] Lebech AM. Polymerase chain reaction in diagnosis of Borrelia burgdorferi infections and studies on taxonomic classification. APMIS Suppl. 2002;(105):1-40. PMID: 11985118 [8] Could Your Dog Have Lyme Disease? How to Recognize the Symptoms and Get Treatment, Lavanya Sunkara , July 2022, GoodRx Health [9] Everything You Need To Know About Lyme Disease In Dogs, Kimberly Alt, July 2022, Canine Journal [10] Lyme Disease (Lyme Borreliosis) in Dogs, Reinhard K. Straubinger, DrMedVetHabil, PhD, Institute for Infectious Diseases and Zoonoses, Department of Veterinary Sciences, Faculty of Veterinary Medicine, LMU, October 2022 [11] Lyme disease: A pet owner’s guide, American Veterinary Medical Association



