Bartonella henselae: An Infectious Pathogen among Cats
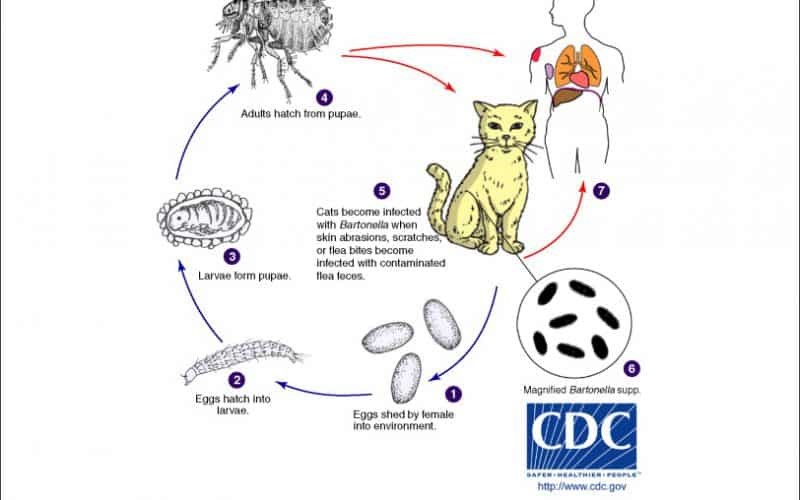
Table of Contents Maigan Espinili Maruquin 1. Introduction Overview of Bartonella henselae as a Significant Feline and Zoonotic Pathogen Bartonella henselae is a small, fastidious, Gram-negative, facultative intracellular bacterium with a global distribution. It exhibits a marked tropism for endothelial cells and erythrocytes, enabling the establishment of chronic, relapsing bacteremia that may persist for months or even years in infected hosts. Domestic cats are the primary mammalian reservoir and represent the principal source of zoonotic transmission to humans. Kittens and feral cats typically harbor higher bacterial loads, although subclinical infection is widespread across the global feline population. Reported bacteremia prevalence in apparently healthy cats ranges from 8% to 56%, depending on geographic region and flea exposure. The primary competent vector for B. henselae is the cat flea (Ctenocephalides felis), within which the organism replicates in the flea gut and is excreted in flea feces (commonly referred to as flea dirt). These contaminated feces can remain infectious in the environment for at least nine days, facilitating indirect transmission. Importance in Companion Animal Medicine and Public Health From a companion animal medicine perspective, B. henselae is clinically significant because most infected cats function as asymptomatic carriers, silently sustaining zoonotic risk. Nevertheless, increasing evidence links B. henselae infection to sporadic but severe feline disease manifestations, including endocarditis, myocarditis, and ocular inflammatory conditions such as uveitis. In dogs, which are considered accidental hosts, bartonellosis is often more pathogenic and has been strongly associated with culture-negative endocarditis and granulomatous inflammatory disease. In public health, B. henselae is best known as the primary etiological agent of Cat Scratch Disease (CSD) in humans. Transmission most commonly occurs when cat claws or oral cavities become contaminated with infected flea feces, which are then inoculated into human skin through a scratch or bite. Immunocompetent individuals typically develop a self-limiting illness characterized by regional lymphadenopathy, fever, and a papule at the site of inoculation. Immunocompromised individuals, including those with HIV/AIDS or organ transplant recipients, are at risk for severe and potentially fatal complications, such as bacillary angiomatosis, bacillary peliosis, encephalitis, and endocarditis, reflecting the organism’s vasoproliferative potential. Scope of the Review and Relevance to Clinical Practice A clear understanding of the epidemiology, pathogenesis, and persistence mechanisms of B. henselae is essential for effective clinical management and disease prevention. Diagnosis remains particularly challenging, as the organism is highly fastidious and slow-growing, frequently resulting in false-negative blood culture findings and so-called “culture-negative” infections. Although molecular assays such as PCR and serological testing are widely employed, interpretation is complicated by intermittent bacteremia and the high background seroprevalence among healthy cats. In clinical practice, adoption of a One Health framework is critical. Veterinarians play a central role in mitigating zoonotic risk through owner education, emphasizing strict, year-round flea control, appropriate hygiene, and cautious interaction with cats, especially in households containing immunocompromised individuals. Management is further complicated by the absence of a standardized antimicrobial protocol capable of reliably achieving complete bacteriological clearance in feline hosts. To conceptualize its biological behavior, B. henselae may be likened to a “stealthy hitchhiker.” By residing within erythrocytes and vascular endothelium, the organism evades immune surveillance, periodically re-emerging only to secure transmission via a passing flea. 2. Characteristics and Epidemiology 2.1 Taxonomy and Microbiological Characteristics The genus Bartonella comprises small, thin, fastidious, and pleomorphic Gram-negative bacilli. These organisms are facultative intracellular pathogens with a highly specialized biological niche characterized by a pronounced tropism for endothelial cells and erythrocytes (red blood cells). Following host entry, Bartonella spp. proliferate within membrane-bound vacuoles, often referred to as invasomes, inside vascular endothelial cells. Periodic release into the bloodstream allows subsequent invasion of erythrocytes, within which the bacteria may persist until cellular senescence or destruction occurs (Cunningham and Koehler 2000; LeBoit 1997). Transmission is predominantly arthropod-borne, involving vectors such as fleas (Ctenocephalides felis), ticks, lice, and sand flies. Although Bartonella species have been isolated from a wide range of mammalian hosts, including rodents, rabbits, canids, and ruminants, domestic and feral cats represent the principal mammalian reservoir for the most epidemiologically and clinically significant zoonotic species, particularly B. henselae (Chomel et al., 1996; Pennisi, Marsilio et al., 2013; Guptill, 2012). 2.2 Global Distribution and Seroprevalence Bartonella species exhibit a global distribution, although prevalence varies markedly according to environmental and ecological conditions. In European feline populations, reported antibody prevalence ranges from 8% to 53% (Pennisi, Marsilio et al., 2013; Zangwill, 2013), while global serological evidence of exposure in cats spans approximately 5% to 80% (Guptill, 2012). The epidemiology of Bartonella infection is strongly influenced by geography, climate, and flea density. The highest prevalence rates are consistently observed in warm, humid temperate and tropical regions, where environmental conditions favor the survival and propagation of C. felis. In contrast, in colder climates, such as Norway, Bartonella infection in cats is reported to be rare or virtually absent, reflecting the limited persistence of flea vectors under such conditions. 2.3 Species Diversity and Genotypes Although 22 to 38 Bartonella species have been described to date, Bartonella henselae remains the most frequently detected species in both domestic cats and humans. Feline populations may also harbor Bartonella clarridgeiae, identified in approximately 10% of infected cats, while B. koehlerae is detected far less commonly (Guptill, 2012). Considerable regional variation exists among B. henselae genotypes, which are broadly classified into Houston-1 (Type I) and Marseille (Type II) strains. Type II (Marseille) predominates among feline populations in the western United States, western continental Europe, the United Kingdom, and Australia. Type I (Houston-1) is the dominant genotype in Asia, including Japan and the Philippines, and is most frequently isolated from human clinical cases worldwide, even in regions where Type II strains are more prevalent among cats. Beyond domestic cats, Bartonella infections have been documented in non-domestic felids, including African lions, cheetahs, and various neotropical wild cat species, underscoring the broad ecological adaptability of the genus (Guptill, 2012). To conceptualize its ecological behavior, Bartonella may be viewed as a “weather-dependent squatter.” It establishes itself most successfully in warm, densely populated environments rich in flea vectors,
Feline Pancreatic Lipase (fPL)
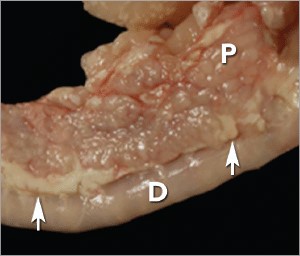
Andy Pachikerl, Ph.D Introduction Pancreatitis appears to be a common disease in cats,1 yet it remains frustratingly difficult to establish a clinical diagnosis with certainty. Clinicians must rely on a combination of compatible clinical findings, serum feline pancreatic lipase (fPL) measurement, and ultrasonographic changes in the pancreas to make an antemortem diagnosis, yet each of these 3 components has limitations. Acute Versus Chronic Pancreatitis Acute pancreatitis is characterized by neutrophilic inflammation, with variable amounts of pancreatic acinar cell and peripancreatic fat necrosis (Figure 1).1 Evidence is mounting that chronic pancreatitis is more common than the acute form, but sonographic and other clinical findings overlap considerably between the 2 forms of disease.1-3 Diagnostic Challenges Use of histopathology as the gold standard for diagnosis has recently been questioned because of the potential for histologic ambiguity.3,4 A seminal paper exploring the prevalence and distribution of feline pancreatic pathologic abnormalities reported that 45% of cats that were apparently healthy at time of death had histologic evidence of pancreatitis.1 The 41 cats in this group included cats with no history of disease that died of trauma, and cats from clinical studies that did not undergo any treatment (control animals). Conversely, multifocal distribution of inflammatory lesions was common in this study, raising the concern that lesions could be missed on biopsy or even necropsy. Prevalence Such considerations help explain the wide range in the reported prevalence of feline pancreatitis, from 0.6% to 67%.3 The prevalence of clinically relevant pancreatitis undoubtedly lies somewhere in between, with acute and chronic pancreatitis suggested to represent opposite points on a disease continuum.2 FIGURE 1. Duodenum (D) and duodenal limb of the pancreas (P) in a cat with acute pancreatitis and necrosis; well-demarcated areas of necrosis are present at the periphery of the pancreas in the peripancreatic adipose tissue(arrows). Courtesy Dr. Arno Wuenschmann, Minnesota Veterinary Diagnostic Laboratory Risk factors No age, sex, or breed predisposition has been recognized in cats with acute pancreatitis, and no relationship has been established with body condition score.3-5 Cats over a wide age range, from kittens to geriatric cats, are affected; cats older than 7 years predominate. In most cases, an underlying cause or instigating event cannot be determined, leading to classification as idiopathic.3 Abdominal trauma, sometimes from high-rise syndrome, is an uncommon cause that is readily identified from the history.6 The pancreas is sensitive to hypotension and ischemia; every effort must be taken to avoid hypotensive episodes under anesthesia. Comorbidities In cats with acute pancreatitis, the frequency of concurrent diseases is as high as 83% (Table 1).2 Pancreatitis complicates the management of some diabetic cats and may induce, for example, diabetic ketoacidosis.7 Anorexia attributable to pancreatitis can be the precipitating cause of hepatic lipidosis.8 The role of intercurrent inflammation in the biliary tract or intestine (also called triaditis) in the pathogenesis of pancreatitis is still uncertain. Roles of Bacteria In one study, culture-independent methods to identify bacteria in sections of the pancreas from cats with pancreatitis detected bacteria in 35% of cases.9 This report renewed speculation about the role of bacteria in the pathogenesis of acute pancreatitis, and the potential role that the common insertion of the pancreatic duct and common bile duct into the duodenal papilla may play in facilitating reflux of enteric bacteria into the “common channel” in cats. Awareness of triaditis may affect the diagnostic evaluation of individual patients. Table 1. Clinical Data from 95 Cats with Acute Pancreatitis (1976—1998; 59% Mortality Rate) & 89 Cats Diagnosed with Acute Pancreatitis (2004—2011; 16% Mortality Rate) PARAMETER HISTORICAL DATA* CATS WITH PANCREATITIS† SURVIVING CATS WITH PANCREATITIS† Number of Cats 95 89 75 ALP elevation 50% 23% 18% ALT elevation 68% 41% 36% Apparent abdominal pain 25% 30% 32% Cholangitis NA 12% 11% Concurrent disease diagnosed NA 69% 68% Dehydration 92% 37% 42% Diabetic ketoacidosis NA 8% 5% Diabetes mellitus NA 11% 12% Fever 7%‡ 26% 11% GGT elevation NA 21% 18% Hepatic lipidosis NA 20% 19% Hyperbilirubinemia 64% 45% 53% Icterus 64% 6% 6% Vomiting 35%—52% 35% 36% ALP = alkaline phosphatase; ALT = alanine aminotransferase; GGT = gamma glutamyl transferase; NA = not available * Summarized from 4 published case series; a total of 56 cats had acute pancreatitis diagnosed at necropsy and 3 by pancreatic biopsy5,8,10,11 † Data obtained from reference12 ‡ 68% of cats were hypothermic DIAGNOSTIC EVALUATION Many cats with pancreatitis have vague, nonspecific clinical signs, which make diagnosis challenging.5 Clinical signs related to common comorbidities, such as anorexia, lethargy, and vomiting, may overlap with, or initially mask, the signs associated with pancreatic disease. Early publications on the clinical characteristics of acute pancreatitis required necropsy as an inclusion criterion, presumably skewing the spectrum of severity of the reported cases.5,8,10,11 Cats with chronic pancreatitis were excluded from these reports. Clinical Findings Table 1 lists common clinical findings in cats from necropsy-based reports and a recent series of 89 cats with acute pancreatitis studied by the authors.12 Note the lower prevalence of most clinical findings in the cats diagnosed clinically rather than from necropsy records. In our evaluation of affected cats, 17% exhibited no signs aside from lethargy and 62% were anorexic. Vomiting occurs inconsistently (35%—52% of cats). Abdominal pain is detected in a minority of cases even when the index of suspicion of pancreatitis is high. About ¼ of cats with pancreatitis have a palpable abdominal mass that may be misdiagnosed as a lesion of another intra-abdominal structure. Laboratory Analyses Hematologic abnormalities in cats with acute pancreatitis are nonspecific; findings may include nonregenerative anemia, hemoconcentration, leukocytosis, or leukopenia. Serum biochemical profile results vary (Table 1). In our acute pancreatitis case series, 33% of cats had no abnormalities in their chemistry results at presentation.12 Serum cholesterol concentrations may be high in up to 72% of cases. Some cases of acute pancreatitis are associated with severe clinical syndromes, such as shock, disseminated intravascular coagulation, and multiorgan failure, that influence some serum parameters, such as albumin, liver enzymes, and coagulation tests. Plasma ionized calcium concentration may be low, and has
Concurrent with T-zone lymphoma and high-grade gastrointestinal cytotoxic T-cell lymphoma in a dog
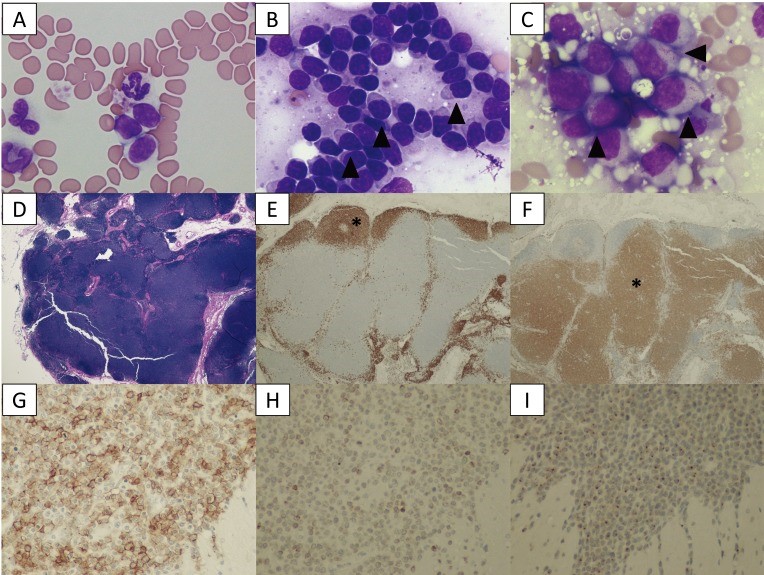
Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5402196/ A 9-year-old, spayed female Golden Retriever dog showed lymphocytosis and lymphadenopathy, secondary to suspected chronic lymphocytic leukemia (CLL). Small-to-intermediate lymphocytes were observed from the cytological examination of the right popliteal lymph node via a fine-needle aspirate. The dog was suspected to have a low-grade lymphoma based on the finding of cytology. Also, ultrasonography reveled thickened lesions in the stomach and small intestine. Histopathology of the popliteal lymph node and small intestine revealed a simultaneous presence of T-zone lymphoma (TZL) and high-grade gastrointestinal (GI) cytotoxic T-cell lymphoma. PCR for antigen receptor rearrangements assay suggested that both lymphomas, though both originated in the T-cells, derived from different genes. The dog died 15 days after diagnosis, despite chemotherapy. Fig. 1. A–C: Cytological images on day 1. (A) Peripheral blood smear. Increased numbers of small lymphocytes. (B) Cytology of the popliteal lymph node biopsy. Most lymphocytes are small-to-intermediate, mature lymphocytes. Some lymphocytes show a “hand mirror” type of cytoplasmic extension (arrowhead) (Wright-Giemsa stain, × 400). (C) Slide preparation of tissue from the small intestine. The lymphocytes are intermediate-to-large, immature cells, and some display azurophilic granules in the cytoplasm (LGLs, arrowhead). D–F: Histological images of popliteal lymph node tissue. (D) Hematoxylin and eosin (H&E) staining. (E) The lymphocytes with fading follicular structures are CD20 positive (asterisk). Immunolabeling with anti-CD20, a hematoxylin counterstain. (F) The nodal capsule (CD3 positive) is thinned without the involvement of the perinodal tissue (asterisk). Immunolabeling with anti-CD3, hematoxylin counterstain. G–I: Histological images of the intestinal tissue. All lymphocytes are positive for CD20 (G), CD3 (H) and granzyme B (I). Fig. 2. (A) Transverse ultrasound image on day 1 showing a thickened intestinal wall (approximately 9.0 mm, arrowhead). (B) Post-contrast transverse CT image on day 2 also showing a thickened intestinal wall (arrowheads). The intrathoracic and abdominal lymph nodes are enlarged. Fig. 3. PARR analysis. (A) The peripheral blood sample shows TCRγ gene rearrangement. (B) The intestinal tissue sample also shows TCRγ gene rearrangement. The two tumors demonstrate clonal expansions from different primers.
